Aquaculture Activities Regulations guidance document
Table of Contents
- Purpose
- Background
- Scope of Application
- Aquaculture Monitoring Standard
- Implementation
- Consequences of Non-Compliance
- Contact Us
- Section-by-Section Guidance
- Section 1: Definitions
- Section 2: Specified substances
- Section 3: Conditions applicable to deposits
- Section 4: Aquaculture facility
- Section 5: Drugs
- Section 6: Pest control products
- Section 7: Measures to reduce detriment
- Section 8: Information required before depositing
- Section 9: Applications affecting contours of footprint
- Section 10: Substrate sampling and restocking
- Section 11: Visual monitoring of substrate; monitoring and restocking
- Section 12: Notification of Minister
- Section 13: Fish morbidity or mortality
- Section 14: Annual report
- Section 15: Prescribed works, undertakings, activities and conditions
- Section 16: Annual report
- Section 17: Paragraph 35(2)(a) of Act
- Section 18: Registration
- Annex 1: AAR Reporting Template
- Annex 2: AAR Application
- Annex 3: Regional Aquaculture Management Offices
- Annex 4: Reporting a morbidity/mortality event
- Annex 5: Drugs and Pesticides Requiring Testing under the AAR (s.13)
- Annex 6: Sampling Guidance for Drugs and Pest Control Products
- Annex 7: Monitoring Standard
- Annex 8: Program Protocols for Marine Finfish Environmental Monitoring in British Columbia
- Acronyms, Abbreviations and Definitions
- 1. Compliance Sampling Locations
- 2. Benthic Monitoring and Reporting Requirements
- 3. Video Surveys
- 4. Sediment Sampling
- 5. Quality Assurance / Quality Control of Sediment Samples
- 6. Calibrating the Sulphide (Ag+/S=) Electrode
- 7. Standardizing the Redox (Pt) Electrode
- 8. Performing Statistical Analyses
- Annex 9: Monitoring Protocol for Benthic Substrates under Marine Finfish Farms in Newfoundland and Labrador
- Annex 10: Summary of required actions
- Annex 11: Glossary of terms
Guidance Document for Owners and Operators
Disclaimer
The Aquaculture Activities Regulations Guidance Document is for information only. It is not a substitute for the Fisheries Act or its Regulations. In the event of an inconsistency between the Guidance Document and the Fisheries Act or its Regulations, the legislation always prevails. In addition, the Aquaculture Activities Regulations do not replace or repeal any existing federal or provincial statutes.
Effective Date and Review
The Aquaculture Activities Regulations Guidance Document is effective on the day on which the Regulations come into force. The Guidance Document will be updated, as needed, to provide further clarity as the Regulations are implemented. The most recent version of this document, together with other Aquaculture Activities Regulations-related documents, can be found on the Fisheries and Oceans Canada website.
Purpose
The purpose of the Guidance Document for Owners and Operators is to provide a clear interpretation of what the Aquaculture Activities Regulations (AAR) mean, how they will be implemented, and what owners and operators need to do to comply with their provisions. In addition to the Regulations and this Guidance Document, the following instruments will support AAR implementation: * An Aquaculture Monitoring Standard (Annex 7) (Monitoring Standard), referenced in the Regulations and therefore required to be followed; and * An AAR Reporting Template (Annex 1) for licence holders; while it is not mandatory that this template be used, it provides a convenient checklist for all AAR reporting requirements.
All of these AAR related documents are available here.
Background
In Canada, aquaculture is managed by different levels of government. Provincial governments are the primary regulators and leasing authorities for aquaculture (except in British Columbia and Prince Edward Island), while the federal government has responsibility for navigation, disease prevention affecting international trade, and the environment under the Fisheries Actand the Health of Animals Act. On November 25,2013, amendments were made to the Fisheries Act to focus the habitat protection provision (s.35) on threats to the productivity of recreational , commercial and Aboriginal fisheries. The pollution prevention provision (s.36), which prohibits the deposit of deleterious substances into waters frequented by fish unless permitted by regulation, remained unchanged. In March 2014, a clear delineation of responsibilities for managing threats from deleterious substances (under s.36) between Environment Canada and Fisheries and Oceans Canada (DFO) was established by an Order in Council (known as the s.36 designation order). Under the designation order, the Minister of Environment is the lead minister for all matters related to the deposit of deleterious substances except in relation to the use of such substances for the purpose of aquaculture management and the management of aquatic invasive species and aquatic pests. In these areas, the Minister of Fisheries and Oceans has the lead role. The Aquaculture Activities Regulations (AAR) are made pursuant to the Ministerial regulation-making authorities as stated in sections 35(3) and 36(5.2) of the Fisheries Act. These risk-based Regulations were developed to clarify legalities with respect to the normal operations of an aquaculture facility : those activities related to the structure and maintenance of the facility itself, and those designed to protect farmed fish from pests and pathogens entering from the marine environment and to control the transfer of those pests and pathogens back to wild fish. The three classes of substances managed under the AAR [drugs, pesticides and biochemical oxygen-demanding matter (BOD)] are already managed, for the most part, by provincial and other federal authorities. Some provinces with marine finfish aquaculture have already established BOD limits to minimize impacts from organic deposits. Ontario has developed “Sediment Quality Objectives, Monitoring and Management Response” as conditions of licence for freshwater finfish sites. In addition, provinces and Health Canada have complementary responsibilities for managing how drugs and pesticides are used. The AAR brings together all these pieces of provincial and federal legislation for the greater protection of fish and fish habitat and to bring aquaculture into compliance with the spirit and intent of the Fisheries Act.
Scope of Application
- Application to Fish Farmers
The AAR apply to all aquaculture facilities in Canada which have a provincial or federal licence and whose operations have the potential to deposit deleterious substances into fish -bearing waters or to cause serious harm to fish that are part of a commercial , recreational or Aboriginal fishery or fish that support those fisheries. The Regulations apply to marine and freshwater finfish (including hatcheries), and shellfish; however, sections 8-12 relate only to facilities located in tidal waters that cultivate finfish. Lobster tanks or pounds, herring weirs, or other holding facilities for wild-capture species are not covered or affected by these Regulations. The purpose of the Regulations is to provide authorization, under certain conditions, to all affected facilities to conduct two types of activities: * the deposit of deleterious substances (s.36 Fisheries Act). These owner or operators are authorized to deposit only those drugs permitted for sale under the Food and Drugs Act, only those pest control products registered or authorized under the Pest Control Products Act, and only biochemical oxygen demanding matter (e.g., fish food, feces, biofouling material) related to aquaculture fish production. Owners and operators may deposit drugs and pest control products only after they have determined that these are the most suitable and effective means of treating a fish pest or pathogen, and that failure to use these treatments would result in negative effects for fish health and disease transfer. * operational activities that may cause serious harm to fish that are part of a commercial, recreational or Aboriginal fishery or to species that support those fisheries (s.35 Fisheries Act). These activities are authorized only if the facility is licensed, and only if the owner or operator of the facility takes measures to minimize serious harm to those fish and their habitat.
Since the Regulations are risk-based, not all the requirements stipulated in the AAR will apply to every aquaculture site and not all owners or operators will have to report on every provision. The table in Annex 2 provides an overview of the scope of application and indicates which sections of the Regulations apply to what type of aquaculture facility. These requirements are also detailed in the AAR Reporting Template (in Annex 1). The Regulations do not replace or repeal any existing federal or provincial statutes. The Regulations do not regulate the approval, application, or any other element of drug or pesticide use, or the disposal of fish waste from fish processing activities. DFO will not issue any licences or permits under the AAR; rather, the Regulations constitute the authorization and set conditions for compliance with s.35 and s.36 of the Act. The AAR will not affect the manner in which aquaculture facilities are currently licensed in any province or territory or by DFO in British Columbia and Prince Edward Island. The definition of “aquaculture licence ” in the Regulations reflects the status quo. However, aquaculture facilities that do not have an aquaculture licence (which may be provincial or federal and includes leases which set conditions of operations) are not covered by the AAR and thus are subject to the s.35 and s.36 prohibitions and enforcement provisions of the Fisheries Act at all times. While DFO is responsible for enforcing the AAR, the Department has worked closely with other federal and provincial authorities in the development of the Regulations to avoid regulatory overlap or contradiction. The Regulations align with programs and regulatory regimes already existing in provincial and other federal jurisdictions so as to minimize duplicative administrative requirements, while ensuring that legislated environmental protection objectives are met. The mitigation and monitoring requirements under these Regulations codify, as much as possible, industry requirements and practices already existing under relevant provincial and federal regulatory regimes, while introducing new and cost-effective measures to allow the federal government to better coordinate the management of pesticides and drugs used to treat pests and pathogens at aquaculture sites. * Application to the Canadian Food Inspection Agency
The Regulations also apply to the Canadian Food Inspection Agency (CFIA) whereby the President of CFIA is authorized to deposit deleterious substances and to kill fish for the purposes of implementing the Health of the Animals ActFisheries Act s.35 and s.36 perspective. * Application to Siting of Aquaculture Facilities
Currently, the AAR only require information that would support siting decisions, where requested by provinces, at marine finfish sites. For all other types of facilities, DFO would continue to work with provinces through existing processes (joint site application requirements) to obtain any necessary information that DFO would need to make assessments of potential threats to fish or fish habitat (including species protected by the Species At Risk Act). Either through the AAR or other information-gathering processes, DFO would continue to provide recommendations to provinces to support siting decisions, using the Regional Aquaculture Management Offices (RAMOs) as the single window to the provinces. Except in British Columbia, DFO’s Fisheries Protection and Species At Risk programs will continue to be involved in the siting of aquaculture facilities and will continue to review applications and provide the conclusions of the reviews to licensing authorities, via the RAMOs These Regulations do not change the responsibilities and obligations of owners and operators under the Species at Risk Act. * Science Review
As knowledge and technology advances, other controls or modifications to existing mitigation measures and monitoring protocols may be needed. To that end, DFO and federal partners have committed to undertake a three-year Science Review to support ongoing implementation of the AAR. The DFO commitment under the Science Review will be coordinated by Aquaculture, Biotechnology and Aquatic Animal Health Science Branch in Ottawa, and include the expertise of DFO’s National Contaminates Advisory Group. Core objectives of the Science Review include a review of existing BOD matter monitoring approaches for freshwater and hard-bottom marine finfish sites and post-deposit drug and pesticide monitoring approaches. Science advice will also be provided through DFO’s Canadian Science Advisory Secretariat, and the peer-reviewed scientific analyses and advice will be published on DFO’s website. The Department will consider recommendations for modifying the Regulations and/or the Monitoring Standard that come out of these scientific peer review processes, as appropriate.
Aquaculture Monitoring Standard
The Aquaculture Monitoring Standard (Annex 7) (Monitoring Standard) is a document incorporated by reference into the Aquaculture Activities Regulations. "Incorporation by Reference" is a term used to describe a mechanism which allows a document that is not in the text of the regulations to be made a part of the regulations. One way to do this would be to reproduce the text of the incorporated document word for word in the regulations. Alternatively, the title of the document can simply be referred to, making the contents of that document “incorporated by reference.” The legal effect of incorporation by reference is to write the words of the incorporated document into the regulations just as if the document had been reproduced word for word. The Monitoring Standard is referenced in sections 8, 9, 10 and 11 of the AAR. In order to be in compliance with the Regulations, owners and operators of marine finfish facilities must use the procedures described in the Monitoring Standard to conduct the activities described in these sections. Further guidance on BOD monitoring required by DFO in the provinces of British Columbia and Newfoundland and Labrador can be found in the protocols in annexes 8 and 9, respectively. Guidance on BOD monitoring in other provinces or territories may be found in the conditions of licence or other requirements specific to those jurisdictions.
Implementation
Within DFO, the AAR will be implemented primarily by the Aquaculture Management Directorate, the Regional Aquaculture Management Offices, with support from Conservation and Protection, Fisheries Protection Program, Species at Risk Program and Ecosystems and Oceans Science, as follows: Regional Aquaculture Management Offices (RAMOs) * The RAMOs will be the key contact for industry interaction; * They are the intake point for notifications [s.6(d), the reports required by s.8 and s.9, exceedances of BOD thresholds as per s. 12(3)and the annual reports in s.16]; RAMOs will liaise with Conservation and Protection (C&P), Fisheries Protection Program (FPP), Ecosystems and Oceans Science (EOS) to determine morbidity / mortality and sampling requirements [s.13(2)]; * Each RAMO may develop a list of aquaculture sites in the region, along with names and contact information of their owners or operators. This list can be used to determine any delinquencies in annual reporting; * RAMOs will forward questions from owners and operators regarding the Regulations, the Guidance Document for Owners and Operators, and the Monitoring Standard to AMD Headquarters for interpretation.
Aquaculture Management Directorate (AMD): National Oversight * AMD will maintain national consistency for interpretation and implementation through updates to the Guidance Document; * AMD will develop and manage the reporting database ; and * AMD is responsible for preparation of all public reports.
Conservation and Protection (C &P) * C&P will work with Environment Canada to ensure appropriate and comprehensive transfer of compliance and enforcement knowledge during the three-year transition period agreed to by the two Departments ; * C&P will be the point of entry for owners or operators to report morbidity /mortality events [s.13(1)], and in consultation with the RAMO and other experts, will provide instructions on required sampling [s.13(2)] according to the Monitoring Standard; * If required, and in consultation with the RAMO, the fishery officer may follow up with owners and operators who are delinquent in filing their annual reports as required in s.16; * If required, and in consultation with the RAMO where appropriate, compliance and enforcement actions such as farm-site inspections, investigations, warnings and/or charges for non-compliance could be taken, and annual inspections of aquaculture sites, based on the overall Departmental priorities for C&P may also be conducted to verify compliance with the AAR; and * Potential enforcement actions could be triggered either directly under the AAR or under the Fisheries Act s.35, 36, and 49.
Fisheries Protection Program (FPP), Species at Risk Program (SRP) and Fisheries Management (FM) * Fisheries Protection Program (FPP), Species at Risk Program (SRP) and Fisheries Management (FM) will provide input as requested on marine aquaculture siting, based on data obtained through the AAR (sections 8 and 9), to the RAMO. For all other types of aquaculture facilities , the RAMO may request input from the other relevant DFO groups on data submitted via the typical provincial licencing/leasing application process. * Respecting s.35 provisions of the AAR, FPP may provide additional expert advice on data interpretation to the RAMO.
Ecosystems and Oceans Science (EOS) * Aquaculture, Biotechnology and Aquatic Animal Health Science, with the expertise found within the Environment and Biodiversity Science Branch’s National Contaminants Advisory Group, will lead the development and implementation of the Science Review to look at post-deposit monitoring of drugs and pest control products ; and * Provide science advice on BOD matter impacts and monitoring protocols for finfish aquaculture located in marine environments where sediment sampling cannot reliably be obtained, and freshwater environments. * Provide science advice on the procedures to be followed for the collection of wild fish carcasses for chemical analyses of the active ingredients found in anti-sea lice chemotherapeutant products. * Science (Regional) will provide input and science advice on marine aquaculture siting. This input will be part of the advice DFO gives to the province as the Department’s contribution to provincial siting decisions for new aquaculture sites. * National and Regional EOS to be involved in the post-deposition/treatment triage to determine morbidity / mortality and sampling requirements [s.13(2)].
Consequences of Non-Compliance
If any of the conditions of the AAR are breached, then the s. 35 and s. 36 prohibitions of the Act immediately apply. As a consequence, DFO or cross-designated officials in inland provinces may lay charges, with offences subject to s.40 of the Act. Offences are punishable on summary conviction to a fine of up to $8 million and/or imprisonment up to six months, and on an indictable offence, to a fine of up to $12 million and/or imprisonment up to three years. Any continuing contravention of the AAR constitutes a separate offence for each day on which the contravention is committed. Canadians are encouraged at all times to report fisheries violations when they observe them. In any province or territory, this may be done by calling Crime Stoppers at 1-800-222-TIPS (8477) or by calling the provincial numbers found on the DFO web site.
Contact Us
Users of this Guidance Document are invited to submit questions and comments to their Regional Aquaculture Management Office (see Annex 3 for contact information). As the AAR are implemented, further changes may be required, and important clarifications may be issued at any time. The Guidance Document will be formally reviewed annually; however, the most recent version of the Guidance Document will be found on the DFO web site.
Section-by-Section Guidance
Section 1: Definitions
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Interpretation | |
| 1. The following definitions apply in these Regulations. | |
| Act : means the Fisheries Act. | |
| Aquaculture : means the cultivation of fish. | |
| Aquaculture licence : means any of the following: | |
| (a) a lease or licence issued or granted by the Minister under section 7 or 58 of the Act, or its regulations, for the purpose of aquaculture; | |
| (b) a lease granted by the government of a province under subsection 59(1) of the Act for the cultivation of oysters; | |
| (c) a provincial licence or authorization for the operation of an aquaculture facility. | |
| Barren substrate: means benthic substrate on which there are no visible organisms. | |
| Biochemical oxygen demanding matter: means any organic matter that contributes to the consumption of oxygen that is dissolved in water or sediment. | |
| Monitoring Standard : means the Aquaculture Monitoring Standard, as amended from time to time, that is produced by the Minister and maintained on the Department of Fisheries and Oceans website. | |
| Soft bottom: means a benthic substrate that consists of loose particles such as clay, mud, marl, sand, pebbles, gravel, shells or small stones. | A number of terms are not defined in the Regulations for one of several reasons: |
| * The term is already defined by a common dictionary meaning (e.g., “reasonable” defined as having sound judgment; fair and sensible); | |
| * The term is already defined under the Act or another federal statute or regulation (e.g., serious harm to fish , pest); | |
| * A definition in regulations is not feasible as there is too much variability in interpretation for inclusion in a legal instrument. |
This Guidance Document provides direction on how many of these terms should be interpreted. Words in bold and italics can be found in the Glossary of Terms inAnnex 11.
Regarding the definition of aquaculture licence(c), “authorization” is intended to cover various terms that could be used (e.g., “permit”) to signify that a facility was authorized to operate by the responsible provincial governing body.
The AAR apply to all licensed aquaculture operations in Canada – marine and freshwater finfish (including hatcheries), and shellfish – that have the potential to cause serious harm to fish which are part of or support a commercial , recreational or Aboriginal fishery, or that may deposit deleterious substances into fish-bearing waters. |
Section 2: Specified substances
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Deleterious Substances | |
| 2. For the purpose of paragraph 36(4)(c) of the Act, the following classes of substances deposited in the operation of an aquaculture facility are specified to be deleterious substances: | |
| (a) drugs whose sale is permitted or otherwise authorized, or whose importation is not prohibited, under the Food and Drugs Act; | |
| (b) pest control products that are registered, or whose use is authorized, under the Pest Control Products Act; and | |
| (c) biochemical oxygen demanding matter. | Intent: |
| The purpose of this section is to define the specific classes of deleterious substances that will be authorized for deposition under these Regulations. Guidance: | |
| In practical terms, these are substances that could, in certain concentrations, directly cause death or produce sub-lethal effects (e.g. tissue damage) to an extent that the sustainability of the fish population in question is threatened. | |
| Substances that may be used to treat pests at aquaculture sites (e.g., lime, brine, and various disinfectants), which are not registered as drugs or pesticides , are not included in the AAR. The normal use of these substances at aquaculture sites is at concentrations that are rapidly diluted in the ocean, mitigating any potential deleterious effects to neighbouring fish and surrounding fish habitat. | |
| The deposition of other, non-prescribed deleterious substances, such as oil or heavy metals, are not authorized under the AAR and are therefore subject to s.36 prohibitions under the Act. | |
| ## Section 3: Conditions applicable to deposits | |
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
| --- | --- |
| Deposit | |
| 3. An owner or operator of an aquaculture facility may, subject to the conditions set out in sections 4 to 14, deposit a deleterious substance specified in section 2 in any water or place referred to in subsection 36(3) of the Act. | Intent: |
| Section 3 of the AAR authorizes the owner or operator of an aquaculture facility to deposit into water the classes of deleterious substances mentioned in section 2, as long as the conditions outlined in section 4 through 14 are met. | |
| These Regulations do not regulate the approval, application, or any other element of drug or pesticide use (other than what is specified), or the disposal of fish waste from fish processing activities. Guidance: | |
| To be authorized, the deposit of a substance during treatment must be in compliance with other federal laws: | |
| * If the substance is a drug, its sale is in compliance with the federal Food and Drugs Act (FDA), and if it is a prescription drug under the FDA, the drug has been prescribed by a veterinarian; and | |
| * If the substance is a pest control product (pesticide), its use is in compliance with the Pest Control Products Act(PCPA). |
The term “owner or operator” encompasses anyone working for the facility or acting as an agent of the owner or operator, whether full or part-time, casual or contract labour. |
Section 4: Aquaculture facility
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Conditions | |
| 4. The deleterious substance must be deposited in the operation of the aquaculture facility and the facility must be operated under an aquaculture licence. | Intent: |
| This section establishes additional parameters under which the deposition of the three classes of deleterious substances can take place. Guidance: | |
| The deposition of a prescribed deleterious substance is only authorized if | |
| * It is deposited as a result of the direct operation of the aquaculture facility ; and | |
| * The aquaculture facility is licensed under one of the classifications of licence described in s.1 of the AAR. |
An aquaculture facility is defined as the location where aquaculture activities are being conducted and encompasses net pens, walkways, barges, floating structures, tanks, vessels (e.g., well boats), and living accommodations, and any associated lines and anchors. Equipment used in the rearing, handling or transport of fish, or in the operation, cleaning (including off-site net cleaning), disinfection and care of the physical structures themselves (whether on or off lease) is considered to be part of the facility. |
Section 5: Drugs
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Conditions | |
| 5. In the case of a deposit of a drug, | |
| (a) if by or under an Act of Parliament the drug may only be sold under a prescription, it must be prescribed by a person who is duly authorized to practise veterinary medicine | |
| (i) under the laws of the province in which the aquaculture facility is located, or | |
| (ii) under the laws of any province, if the aquaculture facility is not located in a province; | |
| (b) the owner or operator of the facility must take measures to minimize the risk of an accidental deposit of the drug; and | |
| (c) if the drug is deposited to control a pest as defined in the Pest Control Products Act, the owner or operator must consider, before depositing the drug, whether there are alternatives to the deposit of that drug and make a record of that consideration. Refer to: | |
| s.5(b): AAR Reporting Requirement 1 | |
| s.5(c): AAR Reporting Requirement 2 | |
| (see Annex 1) | Intent: |
| Section 5 further articulates conditions under which drugs may be deposited during the operation of a licensed aquaculture facility. If the drug that is to be deposited can only be sold by prescription under the Food and Drugs Act, the drug must be prescribed by an authorized veterinarian. | |
| Only a licensed veterinarian may legally prescribe a drug under conditions that are not listed on the label (extra-label use). Guidance: | |
| Paragraph 5(a)(ii) refers to offshore aquaculture facilities located outside of provincial waters. There are currently no offshore aquaculture facilities in Canada. | |
| Paragraph 5(b) requires that owner or operators take measures to avoid accidental deposits of drugs and to demonstrate that measures have been taken to minimize accidental deposits. Many of these measures are articulated in spill avoidance plans, fish health management plans, pest control management plans and/or emergency spill plans that are required by provinces. Information can also be extracted from Standard Operating Procedures, Best Management Practices, or other relevant documentation. Storing medicated feeds in containment structures to avoid accidental spills is an example of a best practice to minimize accidental release of drugs into waters. | |
| Prior to making the decision to control a pest by depositing drugs, paragraph 5(c) stipulates that owners or operators must consider alternatives and record them. Such records will be incorporated in the required annual report. Since this assessment is completed from a subjective, qualitative perspective, the onus is on the owner or operator to thoroughly research what is available in Canada as the accepted industry standard or practice and qualitatively describe this. Possible alternatives to the use of drugs include the following: | |
| * Use of cost-effective commercially available containment technologies to avoid deposits of the drug into fish -bearing waters; | |
| * Use of cost-effective commercially available non-chemical biological treatment technologies; | |
| * Use of cost-effective commercially available treatment technologies to render the drug non-toxic to fish. |
Before deciding to use drugs, owners and operators must also take into consideration other technologies, some of which are still in development, including (but not limited to) : * Mechanical removal of sea lice (e.g., warm water); * Biological filters (e.g., mussels); * Sea lice traps (e.g., light attraction); * Cleaner fish; * Vaccines; * Husbandry modifications (e.g., fish density).
|
Section 6: Pest control products
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Conditions | |
| 6. I n the case of a deposit of a pest control product, | |
| (a) if the pest control product is registered, the owner or operator of the aquaculture facility must use it in compliance with any conditions specified under the Pest Control Products Act, including any conditions relating to the place where it may be used and the quantity and concentration that may be used; | |
| (b) if the pest control product is not registered, it must be authorized to be used by the owner or the operator under subsection 21(5) or 41(1) of the Pest Control Products Act or have been exempted from registration by a regulation made under paragraph 67(1)(z.4) of that Act; | |
| (c) the owner or the operator must consider, before depositing the pest control product, whether there are alternatives to the deposit of that pest control product and make a record of that consideration; and | |
| (d) the owner or the operator must notify the Minister, at least 72 hours before the deposit, of the product name of the pest control product and the time, date and geographic coordinates of the deposit. Refer to: | |
| s.6(c): AAR Reporting Requirement 2 | |
| (see Annex 1) Summary of Required Actions | |
| (see Annex 10) | Intent: |
| Section 6 further articulates conditions under which pest control products may be deposited during the operation of a licensed aquaculture facility. Guidance: | |
| Paragraph 6(a) requires that products registered under the Pest Control Products Act (PCPA) be used according to label conditions. Any deviations from the product label are not only violations under the PCPA, but are now also violations under the AAR. In other words, if the pest control product itself is registered, but if it is used in volumes or concentrations that exceed the label instructions, the user is in violation of both the PCPA and the AAR. | |
| Paragraph 6(b) allows the owner or operator to deposit pest control products that are not registered under the PCPA provided that | |
| * they have been authorized under the PCPA (this is usually done on a case-by-case basis). These substances must meet strict conditions of use as set out by the Pest Management Regulatory Agency (PMRA). | |
| * they are exempted from the PCPA because they pose insignificant risk to environmental or human health. |
All products used to treat sea lice are registered.
Prior to making the decision to control a pest by depositing pest control products, paragraph 6(c) requires the owner or operator to consider alternatives to the use of pest control products and record them as part of the required annual report. Since this assessment is completed from a subjective, qualitative perspective, the onus is on the owner or operator to thoroughly research what is available in Canada as the accepted industry standard or practice and qualitatively describe the practices employed on each site. Possible alternatives to the use of therapeutants include the following :
* Use of cost-effective commercially available containment technologies to avoid deposits of the therapeutant into fish -bearing waters;
* Use of cost-effective commercially available non-chemical biological treatment technologies;
* Use of cost-effective commercially available treatment technologies to render the therapeutant non-toxic to fish.
Paragraph 6(d) requires any owner/ operator planning to deposit a pest control product to notify the Regional Aquaculture Management Office at least 72 hours prior to the deposit of the product. The owner or operator must provide the product name, and time, date and place of deposit. Identification of place should be as specific as possible, using GPS coordinates or their equivalent.
Notification must be made by email. Refer to Annex 3 for Regional Aquaculture Management Offices contact information. Confirmation of receipt of notification will be provided.
The purpose of this provision is to provide the Department with the opportunity to:
* Be aware of when and where depositions will be taking place;
* Be present when the deposition takes place, if the Department deems it necessary; and
* Help inform any investigation that may take place due to a fish kill near an aquaculture facility.
Failure to notify the appropriate officials within the required timeframe constitutes a breach of the conditions of the AAR. When this occurs, the owner or operator is no longer authorized under the AAR and immediately becomes subject to the enforcement provisions of the Fisheries Act (see Consequences of Non-Compliance). If conditions of deposit specific to pesticides are violated, or illegal pesticides are used, then Health Canada’s Pest Management Regulatory Agency may also prosecute under the Pest Control Products Act and the Contraventions Act. |
Section 7: Measures to reduce detriment
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Conditions | |
| 7.(1) The owner or the operator of the aquaculture facility must, in depositing a deleterious substance referred to in paragraph 2(a) or (b), take reasonable measures to minimize detriment to fish and fish habitat outside of the facility, having regard to | |
| (a) the cost and effectiveness of the available measures; | |
| (b) the degree and nature of the detriment that may result from the deposit; and | |
| (c) the physical characteristics of the facility and the type of aquaculture that is engaged in. Refer to | |
| s.7(1): AAR Reporting Requirement 3(a) (Drugs) | |
| s.7(1): AAR Reporting Requirement 3(b) | |
| (Pest Control Products) | |
| (see Annex 1) Feces and unconsumed feed | |
| (2) In the case of an aquaculture facility that cultivates finfish and that is operated under an aquaculture licence that permits a standing biomass of more than 2.5 t or an annual production of more than 5 t, the owner or operator of the facility must take reasonable measures to minimize the deposit of fish feces and unconsumed feed, having regard to the factors set out in paragraphs (1)(a) to (c). Refer to | |
| s.7(2): AAR Reporting Requirement 3 (c) | |
| (see Annex 1) | Intent: |
| In addition to the conditions outlined in sections 5 and 6, section 7(1) requires the owner or operator, when depositing drugs and/or pest control products (as defined in s.2 of these Regulations), to take reasonable actions and/or have reasonable procedures and practices in place to reduce negative impacts to fish and fish habitat in the area outside the aquaculture facility. Guidance: | |
| There are a number of examples of acceptable and reasonable measures that owner or operators can take to minimize detriment to fish and fish habitat from the deposit of drugs and pest control products listed immediately below. Many of these practices are already in place, whether implemented through provincial regulatory requirements or industry Codes of Practice. | |
| As a practical matter, these measures should be effective and cost efficient, they should be appropriate to the scale of the impact that is to be avoided, and they should be appropriate to the limitations of the facility and type of aquaculture. | |
| Section 7(2) requires the owner or operator to take reasonable actions and/or have reasonable procedures and practices in place to reduce negative impacts of feces and unconsumed feed to fish and fish habitat. Measures listed below are examples (not an exhaustive list and not applicable to all types of operations), that would help meet the mitigation requirements of s.7. | |
| Even though s.7(2) focuses on fish feces and feed, owners and operators should also consider mitigation measures for other types of BOD matter not directly covered by the AAR (e.g., blood water, point-source sewage discharges) that may come from the facility. Mitigation measures for these types of BOD matter are included below as guidelines. | |
| The impacts from feces and uneaten feed resulting from an aquaculture facility with a standing biomass less than 2.5 tonne (t) are minimal, if not negligible. | |
| * Examples of measures to minimize detriment from drugs [from the AAR Reporting Template, section 3(a)]: | |
| * Vaccines are used to minimize use of antibiotics or sea lice drugs; | |
| * Mechanical or biological pest controls are used to minimize use of antibiotics or sea lice drugs; | |
| * Handling procedures and stocking densities are used to minimize stress and risks of injury to fish, thereby minimizing the need for drug use and deposit; | |
| * Nutritional supplements and feed products are used to maintain optimum fish health and reduce the use of drugs; | |
| * Procedures are used to minimize the exposure of infected fish to others on-site or at nearby sites by implementing appropriate isolation and containment measures; | |
| * Staff and equipment disinfection procedures are used, employing effective and environmentally friendly products for all gear, personnel, equipment, and working platforms, including boats, when moving between facilities and between containment structures (e.g., net pens, tanks); | |
| * Control measures for predator interactions are used to reduce risk of stress to fish (e.g., feed storage to minimize attracting animals; disposal of excess feed off-site; use of physical exclusion devices), thus minimizing the need for drugs; | |
| * Procedures are used to limit the movement of personnel, contractors, suppliers, and visitors to and between facilities when there is suspicion of, or an actual, infectious disease outbreak; | |
| * Procedures are used to limit the sharing of equipment or gear (e.g., equipment/vehicles, floating structures, nets, containers) between active sites; | |
| * Fallowing of farms is used to support the breaking of disease cycles between successive crops of fish; and/or | |
| * Year-class separation is used (i.e., stocking one year-class of fish on individual farms and in designated bay areas, in order to minimize the intergenerational transfer of pathogens and pests between fish). | |
| * Examples of measures to minimize detriment from pest control products [from the AAR Reporting Template, section 3(b)]: | |
| * Mechanical or biological pest controls are used; | |
| * Pesticide label instructions related to optimum concentrations, delivery, spill avoidance, and re-infection prevention are followed; | |
| * Pesticides are handled by a user certified according to provincial legislation; | |
| * Appropriate procedures are used for storage, disposal, and equipment cleaning to avoid excess deposit of pesticides; | |
| * Pesticide application is timed to minimize exposure to non-target organisms; | |
| * Vaccines are used to minimize use of pest control products ; | |
| * Handling procedures and stocking densities are used to minimize stress and risks of injury to fish, thereby minimizing the need for pesticide use and deposit; | |
| * Nutritional supplements and feed products are used to maintain optimum fish health and reduce the use of pest control products; | |
| * Procedures are used to minimize the exposure of infected fish to others on-site or at nearby sites by implementing appropriate isolation and containment measures; | |
| * Control measures for predator interactions are used to reduce risk of stress to fish (e.g., feed storage to minimize attracting animals; disposal of excess feed off-site; use of physical exclusion devices), thus minimizing the need for pesticides; | |
| * Fallowing of farms is used to support the breaking of disease cycles between successive crops of fish; and/or | |
| * Year-class separation is used (i.e., stocking one year-class of fish on individual farms and in designated bay areas, in order to minimize the intergenerational transfer of pathogens and pests between fish). | |
| * Examples of measures to minimize detriment from feces, unconsumed feed and other BOD matter [from the AAR Reporting Template, section 3(c)]: | |
| * Procedures are in place with respect to feeding methods, feed selection and/or equipment, designed to minimize waste feed and to optimize feed conversion rates without compromising fish health including: | |
| * Use of underwater cameras or hand feeding to observe when feeding by fish has slowed or stopped; | |
| * Use of optimal food to increase feed conversion rates and reduce rate and volume of organic matter released; and | |
| * Feed stored in appropriate containment structures to prevent spillage. | |
| * Provincially required or operator-initiated sediment and/or water quality monitoring programs are in place; | |
| * Procedures should be in place to collect and retain, with minimal leakage, blood generated during harvest and disposal at a licensed processing facility; | |
| * Specific to facilities with a point-source discharge of waste water, facility plans should be in place for the following: | |
| * The screening of point-source outfalls to remove larger, particulate organic matter and reduce the total mass released to aquatic environments; | |
| * Filtration or other forms of effluent treatment to reduce the total mass of released organic and related matter; | |
| * For marine finfish facilities with floating overnight living quarters, BOD matter that is produced by the living quarters and discharged as effluent should be retained in a septic tank designed with a retention time of not less than two (2) days prior to discharge, or other similar measures should be employed, to limit BOD matter discharge, according to relevant federal and provincial regulations. |
Section 8: Information required before depositing
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Conditions | |
| 8.(1) In the case of an aquaculture facility located in tidal waters for the cultivation of finfish that commences operations after the day on which these Regulations come into force, the owner or operator of the facility must submit the following information to the Minister at least 300 days before making a first deposit of a deleterious substance in the operation of the facility: | |
| (a) the predicted contours of the footprint of the biochemical oxygen demanding matter that will be deposited by the facility, calculated in accordance with the Monitoring Standard; | |
| (b) a survey conducted in accordance with the Monitoring Standard that identifies the fish and fish habitat on the seabed that is leased for the operations of the facility and the water column above the seabed; | |
| (c) the bathymetry of the seabed that is leased for the operations of the facility, measured in accordance with the Monitoring Standard; and | |
| (d) in the case of a facility located over a soft bottom, the additional information that is specified in the Monitoring Standard concerning the seabed that is leased for the operations of the facility. | |
| Exception | |
| (2) Despite subsection (1), if an aquaculture facility commences operations during the 300 days after the day on which these Regulations come into force, the owner or operator of the facility must provide the information referred to in paragraphs (1)(a) to (d) within 30 days after the day on which operations were commenced. | |
| Monitoring Standard | |
| (3) The studies undertaken to obtain the information referred to in paragraphs (1)(a) to (d) must be conducted in accordance with the Monitoring Standard. | |
| Non-application | |
| (4) Subsections (1) and (2) do not apply to an aquaculture facility whose aquaculture licence, if issued or granted, permits a maximum standing biomass of 2.5 t or less or a maximum annual production of 5 t or less. Refer to | |
| Aquaculture Monitoring Standard: | |
| Survey for Baseline Information | |
| (see Annex 7) BC Monitoring Protocol | |
| (see Annex 8) NL Monitoring Protocol | |
| (see Annex 9) Summary of Required Actions | |
| (see Annex 10) | Intent: |
| This provision is applicable only to new finfish sites located in tidal waters. In order to accommodate research and experimental facilities, the provision does not apply to operations with a standing biomass of 2.5 t or less or an annual production of 5 t or less. Historically, this information has been provided to enable DFO to provide input into provincial siting decisions. Guidance: | |
| The AAR and the Monitoring Standard (Annex 7) identify the level of environmental protection and monitoring to confirm effectiveness that is acceptable to the Department. Most provinces require information of this nature to be provided prior to issuing a site licence to an owner or operator. | |
| The Monitoring Standard is incorporated by reference into the AAR; compliance requires that the activities must be carried out in accordance with the Monitoring Standard. | |
| If owners or operators of new sites have provided the information specified in 8(1)(a) to (d) to the regulating province, providing it to DFO will likely satisfy this requirement. However, if this information has not been provided to the province or if the information provided does not conform to the requirements of the Regulations and the Monitoring Standard, the owner or operator is required to develop the material and provide it to DFO prior to depositing any deleterious substances. Anything less than the content specified in the Regulations produced according to the methodology outlined in the Monitoring Standard would be considered a contravention of the Fisheries Act. | |
| Further guidance on BOD monitoring required by DFO in the provinces of British Columbia (BC) and Newfoundland and Labrador can be found in the protocols in annexes 8 and 9, respectively. | |
| Guidance on BOD monitoring in other provinces or territories may be found in the conditions of licence or other requirements specific to those jurisdictions. | |
| The report of the baseline survey findings must include at a minimum, the following: | |
| * the input parameters of the depositional model used; | |
| * the unedited recorded images; | |
| * the observations recorded from the underwater survey(s); | |
| * a map with the locations of transects and habitat information generated during these transects at an appropriate resolution (i.e., 1:5000 or finer); the bathymetry of the seabed at a resolution of 10 m contours; and the estimated footprint of deposition of BOD matter in 1, 5, and 10 grams of carbon per square metre per day (g C/m2/day) contours; | |
| * the results of sediment sampling for each sampling location. |
The owner or operator must submit the required information to the Regional Aquaculture Management Office (see Annex 3) at least 300 days (10 months) prior to the first deposit of a deleterious substance. This length of time is based on the assumption that most finfish operations will take four to six months from the time the licence is issued to the time fish are actually in the water and depositing BOD material. That leaves four to six months after receipt of the complete application for the various groups within DFO (Fisheries Protection Program, Species At Risk Program, Aquaculture Management, Science) to review the application and provide advice to the province before the licence is issued. No facilities may be stocked prior to the submission of the reports. Information may be submitted by email or by post and confirmation of receipt will be provided.
Note: DFO will continue to provide information to provinces, when requested, on shellfish and freshwater issues outside of the AAR. At this time, the requirement in the AAR to provide baseline data applies only to marine finfish sites; however, it is possible that the AAR will be amended in the future to require the same information from an expanding freshwater industry, provided facilities are licensed. |
Section 9: Applications affecting contours of footprint
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Conditions | |
| 9. If the owner or operator of an aquaculture facility located in tidal waters that cultivates finfish makes an application under federal or provincial law that, if approved, would authorize any activity that is likely to increase the predicted contours of the footprint of the biochemical oxygen demanding matter deposited by the facility, the owner or operator must conduct the studies necessary to obtain the information referred to in paragraphs 8(1)(a) to (d) and submit the information to the Minister within 30 days after the day on which the application was made. | |
| Non-application | |
| (2) Subsection (1) does not apply to an aquaculture facility whose aquaculture licence, if amended, permits a maximum standing biomass of 2.5 t or less or a maximum annual production of 5 t or less. Refer to | |
| Aquaculture Monitoring Standard: | |
| Survey for Baseline Information | |
| (see Annex 7) BC Monitoring Protocol | |
| (see Annex 8) NL Monitoring Protocol | |
| (see Annex 9) Summary of Required Actions | |
| (see Annex 10) | Intent: |
| This provision requires the same information to be provided for marine finfish sites as is required in s.8(1)(a) to (d); however, s.9 refers only to applications that would result in an increase to the BOD footprint of the facility. An increase in the BOD footprint could result from additional fish on the site or from an expansion of the existing site. If the facility is physically moved from one location to another, the process described in s.8 would apply. | |
| Owners or operators of sites whose amended licences allow a maximum standing biomas s of 2.5 t or less or a maximum annual production of 5 t or less are not required to provide this information. Guidance: | |
| The AAR and the Monitoring Standard (Annex 7) identify the level of environmental protection and monitoring to confirm effectiveness that is acceptable to the Department. Most provinces require information of this nature to be provided prior to approving an increase in the quantity of fish cultivated on a site. | |
| The Monitoring Standard is incorporated by reference into the AAR; compliance requires that the activities must be carried out in accordance with the Monitor | |
| If owners or operators have provided the information specified in 8(1)(a) to (d) to the regulating province as part of the application for increase, providing it to DFO will likely satisfy this requirement. However, if this information has not been provided to the province or if the information provided does not conform to the requirements of the Regulations and the Monitoring Standard, the owner or operator is required to develop the material and provide it to DFO. Anything less than the content specified in the Regulations produced according to the methodology outlined in the Monitoring Standard would be considered a contravention of the Fisheries Act. | |
| Further guidance on BOD monitoring required by DFO in the provinces of British Columbia and Newfoundland and Labrador can be found in the protocols in annexes 8 and 9, respectively. Guidance on BOD monitoring in other provinces or territories may be found in the conditions of licence or other requirements specific to those jurisdictions. | |
| Minimum reporting requirements for this information are as follows: | |
| * the input parameters of the depositional model used; | |
| * the unedited recorded images; | |
| * the observations recorded from the underwater survey(s); | |
| * a map with the locations of transects and habitat information generated during these transects at an appropriate resolution (i.e., 1:5000 or finer); the bathymetry of the seabed at a resolution of 10 m contours; and the estimated footprint of deposition of BOD matter in 1, 5, and 10 g C/m2/day contours; | |
| * the results of sediment sampling for each sampling location. |
The owner or operator must submit the information required to the Regional Aquaculture Management Office within 30 days of making the application to the province. Information may be submitted by email or by post. Confirmation of receipt will be provided. |
Section 10: Substrate sampling and restocking
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Conditions | |
| 10.(1) In the case of an aquaculture facility that is located over a soft bottom and cultivates finfish in tidal waters in or adjacent to Quebec, Nova Scotia, New Brunswick, British Columbia, Prince Edward Island or Newfoundland and Labrador, the owner or operator of the facility | |
| (a) must take samples of the benthic substrate in the manner and at the times and locations specified in the Monitoring Standard and determine the concentration of free sulfide in the samples in accordance with that Standard; | |
| (b) must take additional samples of the benthic substrate, in the manner and at the times and locations specified in the Monitoring Standard, if | |
| (i) in the case of a facility located in tidal waters in or adjacent to Quebec, Nova Scotia, New Brunswick, Prince Edward Island or Newfoundland and Labrador, the mean concentration of free sulfide as calculated at the locations specified in the Monitoring Standard exceeds 3000 µM, and | |
| (ii) in the case of a facility located in tidal waters in or adjacent to British Columbia, the mean concentration of free sulfide as calculated at 30 m and 125 m from the structure that contains the cultivated fish exceeds 1300 µM and 700 µM, respectively; and | |
| (c) must not restock the facility if the concentrations of free sulfide exceed the applicable concentration limits set out in paragraph (b). | |
| Visual monitoring instead of sampling | |
| (2) The owner or operator of an aquaculture facility must conduct visual monitoring in accordance with section 11 instead of the sampling described in subsection (1) if the owner or operator cannot obtain, from each of the locations specified in the Monitoring Standard, benthic substrate samples that | |
| (a) contain substrate to a depth of at least 5 cm; and | |
| (b) have a volume of at least 15 ml and undisturbed sediment-water interface. Refer to | |
| s.8: AAR Reporting Requirement 4 | |
| (see Annex 1) Aquaculture Monitoring Standard: Procedures for Monitoring Sediment Free Sulfide | |
| (see Annex 7) BC Monitoring Protocol | |
| (see Annex 8) NL Monitoring Protocol | |
| (see Annex 9) | Intent: |
| For AAR purposes, sediment sulfide concentrations are used to assess the potential impact of organic matter on the benthic environment of soft bottoms. This monitoring approach is common across the country for marine finfish sites over soft-bottom substrates, and is based on research conducted by DFO scientists. While specific thresholds are different (primarily because of different monitoring protocols and due to differences in site-specific substrate composition and physical characteristics, and other factors), the broad management objectives for environmental protection are consistent among West and East Coast jurisdictions. | |
| Monitoring, using the procedures set out in the Monitoring Standard (Annex 7), must be conducted each production cycle. If the applicable sulfide threshold (concentration limit) is exceeded, confirmatory monitoring is required. If the concentration of sediment sulfide continues to be higher than the threshold set for that particular site, measures must be taken to reduce the concentration to below the threshold before fish can be added to the site. | |
| While the AAR acknowledges that soft bottom may be composed of different materials, only samples that are homogeneous, uniform in content and meet certain sampling quality control criteria as specified in the Monitoring Standard will be accepted for comparison against the sulfide threshold. If these criteria cannot be met, the owner or operator will be required to measure BOD impacts by using visual monitoring in accordance with s.11. Guidance: | |
| Sections 10 (1)(a) and (b) specify that the owner or operator of a finfish aquaculture site located over a soft bottom in tidal waters must measure the concentration of free sulfide therein, in accordance with the Monitoring Standard, to determine the oxic state of the benthos in the vicinity of the site. | |
| If specific thresholds are exceeded by the initial samples, paragraph 10(b) requires that the owner or operator resample (according to the Monitoring Standard). For clarity, these thresholds have been articulated for specific provinces within the Regulations. | |
| The owner or operator must submit these results [i.e., the concentrations of free sulfide referred to in subsection 10(1)] in the annual report according to the requirements of the Monitoring Standard. DFO may audit the results at any time. | |
| Paragraph 10(c) establishes a condition that restricts the operator of a marine finfish aquaculture site from restocking as long as the concentrations of free sulfide exceed the applicable concentration limits set out in paragraph 10(b). This means that no restocking can take place until sulfide concentrations return to accepted levels. | |
| If the conditions in s.10(2) cannot be met because it is not possible to obtain sediment samples of sufficient quality to monitor sulfide, the owner or operator must conduct visual monitoring (see Section 11). | |
| The owner or operator must submit the visual monitoring data collected under subsection 10(2) in the annual report. | |
| ## Section 11: Visual monitoring of substrate; monitoring and restocking | |
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
| --- | --- |
| Conditions | |
| Visual monitoring of substrate 11.(1)This section applies to aquaculture facilities that cultivate finfish in the waters referred to in subsection 10(1) but | |
| (a) are not located over a soft bottom; or | |
| (b) are located over a soft bottom In respect of which subsection 10(2) applies. | |
| Monitoring and restocking | |
| (2) The owner or operator of a facility referred to in subsection (1) | |
| (a) must conduct visual monitoring of the benthic substrate in the manner and at the times and locations specified in the Monitoring Standard; | |
| (b) must not, if the facility is located in tidal waters in or adjacent to Quebec, Nova Scotia, New Brunswick, Prince Edward Island or Newfoundland and Labrador, restock the facility if the visual monitoring shows the presence of Beggiatoa species or similar bacteria, marine worms or barren substrate in more than 70% of the locations specified in the Monitoring Standard; and | |
| c) must not, if the facility is located in tidal waters in or adjacent to British Columbia, restock the facility if visual monitoring shows that Beggiatoa species or similar bacteria or marine worms cover | |
| (i) 10% or more of any four segments of substrate specified in the Monitoring Standard that are within 100 m to 124 m from the fish containment structure, or | |
| (ii) 10% or more of | |
| * the two contiguous segments of substrate specified in the Monitoring Standard that are within 116 m to 124 m from the fish containment structure, and | |
| * the two or more contiguous segments of substrate specified in the Monitoring Standard that are within 124 m to 140 m from the fish containment structure. |
Refer to
Aquaculture Monitoring Standard:
Visual Recording Procedures
(see Annex 7) BC Monitoring Protocol
(see Annex 8) NL Monitoring Protocol
(see Annex 9) | Intent:
Section 11 outlines the visual monitoring thresholds for facilities located over substrates (hard, mixed, soft) for which concentrations of free sulfide cannot be determined by the criteria in s.10(a) and (b). The owner or operator must not restock the facility if the visual monitoring thresholds have been exceeded. Guidance:
Procedures for visual monitoring are contained in the Monitoring Standard(Annex 7). Compliance with the AAR requires that the procedures outlined in the Monitoring Standard be followed.
Research indicates that visual imaging can be used as an appropriate primary tool for assessing benthic changes. Studies have also shown that performance-based indicators such as Beggiatoa species (or similar bacteria) or marine worms (e.g., class polychaeta) can serve as biomarkers of aquaculture impact on the benthic environment when it is not possible to obtain bottom grab samples (Canadian Science Advisory Secretariat Report 2014/017).
Beggiatoa species (or similar bacteria) form bacterial mats and occurs at the interface of oxic and anoxic conditions. Marine worms, such as opportunistic annelids of the class polychaeta, are found atop mixed or hard-bottom substrates in response to high organic input. Both are typically associated with elevated sulfide levels and are valid visual indicators of benthic change due to aquaculture. Substrate which is bare (i.e., where there are no visible organisms) as a result of organic enrichment from deposits of aquaculture activity is also an indicator of hypoxic or anoxic conditions. These are not areas that were bare prior to aquaculture activity.
Currently, there are no AAR requirements to monitor BOD deposit from shellfish facilities, freshwater net pens or land-based facilities releasing into fish-bearing waters. There is also at this time no requirement in the AAR to sample the substrate for finfish sites in freshwater; however, if such sampling is a requirement of other jurisdictions or is required under other Fisheries Actauthorities, owners and operators must comply.
These areas will be investigated as part of a Science Review that will inform monitoring approaches of impacts from aquaculture operations, both on the benthic environment and wild fish populations. This research will inform the need for future regulatory amendments. |
Section 12: Notification of Minister
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Conditions | |
| 12.(1) The owner or operator of an aquaculture facility that cultivates finfish that is located in tidal waters in or adjacent to Quebec, Nova Scotia, New Brunswick, Prince Edward Island, or Newfoundland and Labrador must notify the Minister if the following occurs: | |
| (a) the concentration limits set out in subparagraph 10(1)(b)(i) are exceeded; or | |
| (b) Beggiatoa species or similar bacteria, marine worms or barren substrate are present to the extent referred to in paragraph 11(2)(b). | |
| Notification – British Columbia facilities | |
| (2) The owner or operator of an aquaculture facility located in tidal waters in or adjacent to British Columbia must notify the Minister if the following occurs: | |
| (a) the concentration limits set out in subparagraph 10(1)(b)(ii) are exceeded; or | |
| (b) Beggiatoa species or similar bacteria or marine worms are present to the extent referred to in paragraph 11(2)(c). | |
| Time for notification | |
| (3) The owner or operator must notify the Minister within 14 days after the day on which the samples of the substrate were taken or the visual monitoring conducted, as the case may be. Refer to: Summary of Required Actions | |
| (see Annex 10) | Intent: |
| This section requires the owner or operator to notify the Regional Aquaculture Management Office if BOD concentration limits are exceeded or if Beggiatoa species or similar bacteria, marine worms, or barren substrate are present at the levels indicated in the Regulations, whether determined through substrate sampling or visual monitoring. This notification is necessary for DFO auditing purposes. Guidance: | |
| Section 12(3) requires the owner or operator to notify the Regional Aquaculture Management Office within 14 days after the samples referenced in s.10(1)(b)(ii) and 11(2)(c) have been taken. Notification must be provided by email. Confirmation of receipt of notification will be provided. | |
| The owner or operator is not allowed to restock until sampling results return to accepted levels. | |
| These areas will be investigated as part of a Science Review that will inform monitoring approaches of impacts from aquaculture operations, both on the benthic environment and on wild fish populations. This research will inform the need for future regulatory amendments. | |
| ## Section 13: Fish morbidity or mortality | |
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
| --- | --- |
| Conditions | |
| 13.(1) If fish morbidity or mortality outside the aquaculture facility is observed from any part of the facility within 96 hours after the deposit of any drug or pest control product referred to in paragraph 2(a) or (b), the owner or operator of the facility must immediately notify a fishery officer of | |
| (a) the name of the facility and its operator; | |
| (b) the geographic coordinates of the fish observed; | |
| (c) the estimated number and, if known, species of the fish observed; and | |
| (d) the product name of the drug or pest control product deposited and the date of the deposit. | |
| Minister’s directions | |
| (2) If the owner or operator of an aquaculture facility is directed by the Minister to undertake measures under subsection 36(6) of the Act, and the Minister advises the owner or operator that the direction is related to the notification made by them under subsection (1), the owner or the operator must cease depositing the drug or pest control product referred to in subsection (1) until they have complied with the Minister’s directions. Refer to: | |
| s.13(1): AAR Reporting Requirement 5 | |
| (see Annex 1) List of Drugs and Pesticides that may Require Testing | |
| (see Annex 5) Sampling Guidance for Drugs and Pest Control Products | |
| (see Annex 6) Summary of Required Actions | |
| (see Annex 10) | Intent: |
| The purpose of section 13 is to require the owner or operator to notify a fishery officer and take specific actions if a fish kill or morbidity event is suspected to be linked to pesticide or drug treatment deposits. | |
| Section 13 of these Regulations does not remove or repeal the powers available to fishery officers under s.49 of the Fisheries Act to lay charges in the event of a morbidity/mortality occurrence, if there is a determination of illegal use, misuse of a legal product, or if there is a deposit of non-authorized deleterious substances at aquaculture sites. Guidance: | |
| Paragraph 13(1) establishes the procedures to be followed if the owner or operator observes (through visual inspection or by means of underwater cameras) any wild fish morbidity or mortality outside the facility within 96 hours of depositing a drug or pest control product. All morbidity or mortality events must be reported. Following a report, DFO will determine, using a science-based triage process, whether a Fisheries Acts.36(6) directive will be issued. | |
| In the context of an array of net pens, “outside the facility” means outside the rectangle formed by the placement of anchors on the seabed and the corresponding area on the surface: | |
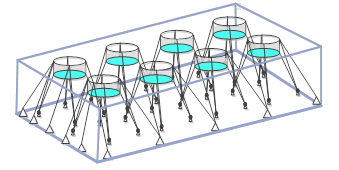 |
|
| The image is a three-dimensional representation of an 8-pen array showing the rectangle outlining the anchors on the bottom and the corresponding outline on the surface of the water. | |
| The owner or operator is required to report any dead or moribund wild fish observed outside this area. Well boats are also deemed to be part of the facility, as are any other physical structures or equipment (see definition of aquaculture facility in Annex 11). | |
| Bear in mind that “owner or operator” encompasses anyone working for the facility or acting as an agent of the owner or operator, whether full or part-time, casual or contract labour. | |
| These procedures include notifying a fishery officer immediately and providing information to the fishery officer concerning location and scale of the morbidity or mortality event, as well as details of the deposition, including name of product and amount deposited. Because of the urgent nature of the notification, placing a call to the appropriate toll-free number (listed in Annex 4) is recommended. Confirmation of receipt will be provided. | |
| If the Department’s triage process indicates that the morbidity/mortality event was caused by circumstances external to the facility and unrelated to the deposit, no further action is required of the owner or operator. These circumstances could include oceanographic conditions (e.g., superchill or algal blooms), disasters (e.g., oil spills), or obvious diseases or viruses affecting wild fish. | |
| However, if the triage process indicates that the morbidity/mortality event may have been caused by the deposit, the Department may instruct the fishery officer to issue a directive under s.36(6) of the Fisheries Act, to “conduct any sampling, analyses, tests, measurements or monitoring, install or operate any equipment or comply with any procedures, and report any information, that is required by the Minister in order to determine whether the person is depositing the deleterious substance in the manner authorized.” | |
| Unless otherwise directed, the owner or operator should follow the procedures contained in Annex 6 in collecting the samples. The fishery officer may be on site to oversee the sampling and retain the samples for testing. If directed by the fishery officer, the owner or operator may be required to send the samples to a laboratory certified by the Standards Council of Canada (see Annex 5 for a list of drugs and pesticides that may require testing). A list of accredited labs will be included in the s.36 directive. The owner or operator is responsible for providing the fishery officer with the name of the laboratory to which the samples have been sent and the date of their expected delivery. | |
| The owner or operator will bear the cost of collecting the samples and sending them to the laboratory (if so directed). DFO will provide advice to the laboratory regarding which tests are to be run and pay for the testing. The testing will focus on active ingredients of authorized products (e.g., emamectin benzoate, SLICE®), as well as illegal products. | |
| Once the owner or operator has complied with all the requirements for sampling, he may resume depositing; however, he should be aware that any continuing contravention of the AAR constitutes a separate offence for each day on which the contravention is committed. | |
| Upon review of all information, the Minister will determine any further action as required. The following are example scenarios. | |
| * The analyses reveal that the cause of the morbidity/mortality event was not related to the deposit of pest control products or drugs. If another type of deposit has occurred within the aquaculture facility, DFO will further investigate. If such deposits occur outside of an aquaculture facility, the responsibility for further investigation is turned over to Environment Canada, as per the s.36 designation order. | |
| * The analyses reveal that the substance was deposited in a legal manner and in legal amounts. The matter is turned over to the Competent Authority (usually Health Canada) which will review the conditions of use and revise such conditions as necessary. The owner or operator may not continue to deposit the substance in question until instructed by the Competent Authority. | |
| * The analyses show that the substance has been deposited in an illegal manner: either the substance itself is illegal or the substance is legally authorized but was not used as authorized. In this case, DFO will investigate further, will notify Health Canada and may take enforcement action. |
Note: The owner or operator is not obligated to report incidents of morbidity or mortality if they occur outside of the 96 hours following deposit of pesticides or drugs. However, as per s.38(5) of the Fisheries Act, owners and operators have a duty to notify DFO or EC if they deposit a deleterious substance not authorized by regulations.
Morbidity/mortality events can also be reported by fishermen or members of the public. Inspections and investigations would be conducted by fishery officers under s.49 of the Act. |
Section 14: Annual report
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Conditions | |
| 14. The owner or operator of an aquaculture facility must submit an annual report to the Minister, and retain a copy of it, in accordance with section 16. | Intent: |
| This section establishes the requirement for the owner or operator to submit an annual report. Guidance: | |
| To assist owners or operators in completing this task, the Department has created the AAR Reporting Template that can be found in Annex 1, or on the DFO website. While use of the template is not mandatory, it is recommended as it provides a checklist for all of the information required for the annual report. | |
| Further guidance is outlined below in s.16 (Annual Report) of the Regulations. | |
| ## Section 15: Prescribed works, undertakings, activities and conditions | |
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
| --- | --- |
| Section 35 of the Act | |
| 15.(1) For the purposes of paragraph 35(2)(a) of the Act, | |
| (a) the following works, undertakings and activities are prescribed: | |
| (i) the installation, operation, maintenance or removal of an aquaculture facility, and | |
| (ii) measures to control biofouling or the presence of fish pathogens or pests in the aquaculture facility; and | |
| (b) the following conditions are prescribed for the carrying on of those works, undertakings and activities: | |
| (i) the aquaculture facility is operated under an aquaculture licence, | |
| (ii) the owner or operator of the facility takes reasonable measures to mitigate the risk of serious harm to fish outside the facility that are part of a commercial, recreational or Aboriginal fishery, having regard to | |
| (A) the effectiveness of the available measures, | |
| (B) the degree of serious harm that may result from the carrying on of the works, undertakings and activities, and | |
| (C) the physical characteristics of the facility and the type of aquaculture that is engaged in at the facility, and | |
| (iii) the owner or operator submits an annual report to the Minister in accordance with section 16. | |
| Exception – deleterious substances | |
| (2) The deposit of deleterious substances is not prescribed for the purpose of paragraph (1)(a). | |
| Exception – Pacific Aquaculture Regulations | |
| (3) Subsection (1) does not apply to any work, undertaking or activity carried on in any waters or place referred to in section 2 of the Pacific Aquaculture Regulations. Refer to | |
| s.15 AAR Reporting Requirement 6 | |
| (see Annex 1) | Intent: |
| Section 35 of the Fisheries Act gives the Minister of Fisheries and Oceans the authority to authorize works, undertakings, and activities as prescribed in 15(1)(a) that cause serious harm to fish, if they are carried out in accordance with the conditions established in 15(1)(b): operating under a licence, taking reasonable measures to minimize serious harm to fish that are part of and support commercial, recreational and Aboriginal fisheries, and submitting an annual report. In British Columbia only, the works, undertakings and activities prescribed and authorized under s.15 of the AAR are instead authorized under the licence conditions of the Pacific Aquaculture Regulations. | |
| Regardless of the authorizing Regulations (i.e., s.15 of the AAR or licence conditions under the Pacific Aquaculture Regulations), the intent is for all licence holders across the country to apply, as a minimum, the same mitigation measures and submit similar information when engaging in activities that may cause serious harm. | |
| Guidance: | |
| The AAR authorizes the regular operations and husbandry activities of an aquaculture facility with respect to controlling biofouling organisms (e.g., through cleaning nets or removing sea lice from fish), and permanently altering or destroying fish habitat(e.g., through the placement of anchors). These activities are only authorized if the conditions described in 15(1)(b) are met. | |
| For example, if serious harm occurs as a result of the activities of an aquaculture facility and DFO determines that insufficient or no measures were taken to minimize that harm (based on the “reasonableness” test), the owner or operator would no longer be authorized, and as a result would be in contravention of s.35. In addition, the onus is on the owner or operator to demonstrate through record-keeping and the AAR annual report that appropriate and sufficient measures were implemented. | |
| Most aquaculture activities are not expected to cause serious harm if precautionary and mitigation measures are implemented. | |
| Mitigation measures minimize impact from aquaculture activities and contribute to protecting fish and fish habitat under this provision. The following measures (not an exhaustive list and not applicable to all types of operations), would help meet the mitigation requirements of s.15. | |
| Note: Although not specifically mentioned in the Regulations, s.15, by implied reference to s.35 of the Fisheries Act, also applies to species that support commercial, recreational and Aboriginal fisheries. | |
| * Examples of measures to minimize serious harm to commercial, recreational and Aboriginal fisheries (from the AAR Reporting Template, section 6)]: |
Measures that could be taken to minimize serious harm to commercial , recreational and Aboriginal fisheries include, but are not limited to the following: * Regular removal of biofouling from infrastructure and nets to ensure that organisms are small when they are removed, thus creating less impact if settling to benthic habitat occurs; * Time work in water to respect timing windows to protect fish, including their eggs, juveniles, spawning adults and/or the organisms upon which they feed; * Restrict type and timing of operations (e.g., shucking, sorting, grading) to minimize release of inorganic matter on the seabed as a result of fish handling or other culture activities; * Design and plan activities and works in water body such that loss or disturbance to aquatic habitat is minimized and sensitive spawning habitats are avoided; * Operate machinery in a manner that minimizes disturbance to the intertidal areas and other fish habitat ; * Avoid locating moorings in important fish habitat and minimize disturbance to submerged aquatic vegetation when securing mooring structures to the seabed; * Consider other relevant measures to avoid causing serious harm to fish and fish habitat; and * For shellfish aquaculture facilities, ensure procedures specific to the species being cultured are in place to minimize the deposit of materials on the substrate.
Section 16: Annual report
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
|---|---|
| Report | |
| 16.(1) An annual report must be submitted in a form acceptable to the Minister and contain the following information in respect of the operation of the aquaculture facility during the calendar year: | |
| (a) for each deposit of a drug or pest control product during the year, | |
| (i) the product name of the drug or pest control product or the common chemical names of its active ingredients, | |
| (ii) the purpose of the deposit, | |
| (iii) the date, quantity and geographic coordinates of the deposit, and | |
| (iv) the record of consideration of alternatives referred to in paragraph 5(c) or 6(c); | |
| (b) a description of any measures taken under paragraph 5(b), section 7 and subparagraph 15(1)(b)(ii); | |
| (c) In the case of a facility referred to in section 10, | |
| (i) the concentrations of free sulfide referred to in subsection 10(1), or | |
| (ii) if visual monitoring is required under subsection 10(2), the visual monitoring data collected; and | |
| (d) if a fishery officer was notified of fish morbidity or mortality in accordance with subsection 13(1), the information provided to the officer. | |
| Due date of report | |
| (2) An annual report must be submitted to the Minister on or before April 1 of the year following the year that is the subject of the report. | |
| Copy of report to be retained | |
| (3) An owner or operator of an aquaculture facility must retain a copy of the annual report in the facility for a period of two years after the day on which it is submitted to the Minister. | |
| Information prior to coming into force | |
| (4) For greater certainty, an owner or operator of an aquaculture facility is not required to include information in an annual report in respect of any period before the day on which these Regulations come into force. Refer to: | |
| s.16 AAR Reporting Requirement 7 | |
| (see Annex 1) Summary of Required Actions | |
| (see Annex 10) | Intent: |
| Section 16 requires that the annual report be submitted in a form acceptable to the Minister, and specifies what information must be reported and by what date. Industry reporting is supported by this Guidance Document and the Aquaculture Monitoring Standard(Annex 7). Guidance: | |
| A suggested Reporting Template is available at Annex 1 and on DFO’s website. While using the reporting template is not mandatory, it is recommended as the template provides a comprehensive checklist of all the information required for the annual report. | |
| Reporting is done on a calendar year basis, with the annual report due April 1, or three months following the end of the year being reported. It is the responsibility of the owner or operator to file a report for each site location. | |
| The completed, signed report is to be submitted to the Regional Aquaculture Management Office (Annex 3) in electronic form or on paper by the date specified in the Regulations. Confirmation of receipt will be provided. | |
| Information provided in the annual reports will be aggregated for the Minister to present to Parliament. As part of the aquaculture public reporting element of the Sustainable Aquaculture Program, an annual report will also be available on the DFO web site. The first annual report – based on industry reports that will be submitted by April 1, 2016, covering the period from July to December, 2015 – will be published in January 2017. | |
| Annual reports must be retained by the owner or operator for a period of two years after submission. | |
| Significant delays in submitting the annual report could result in charges under the Fisheries Act. | |
| ## Section 17: Paragraph 35(2)(a) of Act | |
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
| --- | --- |
| Canadian Food Inspection Agency | |
| 17. (1) For the purposes of paragraph 35(2)(a) of the Act, the disposal of fish by the President of the Canadian Food Inspection Agency for the purposes of fish pathogen or pest control and the Health of Animals Act is prescribed. | |
| Deposit of deleterious substance | |
| (2) The President of the Canadian Food Inspection Agency may, for the purposes of fish pathogen or pest control and the Health of Animals Act, deposit a deleterious substance referred to in paragraph 2(a) or (b) in any water or place referred to in subsection 36(3) of the Act. | Intent: |
| When the Canadian Food Inspection Agency (CFIA) requires treatment to be implemented for fish health management (subject to the Health of Animals Act), some harm may come to fish and fish habitat outside the facility. As a result, CFIA requires authorization for s.35 and s.36-related activities.This section of the AAR provides the necessary authorization. The Agency may ask the owner or operator to carry out the activity on its behalf. | |
| ## Section 18: Registration | |
| Aquaculture Activities Regulations | Guidance for Owners and Operators |
| --- | --- |
| Coming Into Force | |
| 18. These Regulations come into force on the day on which they are registered. | |
| ## Annex 1: AAR Reporting Template | |
 |
|
| ### Annual Report under the Aquaculture Activities Regulations | |
| * NEW Reporting Templates | |
| * Land-Based Operations Template (PDF) | |
| * Marine and Freshwater Cage Finfish Template (PDF) | |
| * Shellfish Template (PDF) | |
| * Additional supporting templates | |
| * Drug and Pesticide Deposit Data Template (Excel) | |
| * Sediment BOD Monitoring Data Template (Excel) | |
| * Visual BOD Monitoring Data Template (Excel) |
Annex 2: AAR Application
| Type of Aquaculture Facility | ||
|---|---|---|
| Open-water Shellfish | Fresh-water Finfish | Open-water Marine Finfish |
| Open-water facilities | Land-based facilities* | |
| AAR Requirements | Section 5: Deposit of Drugs. | |
| 1. Complies with Food and Drugs Act | ||
| 2. Mitigation measures to minimize accidental deposits | ||
| 3. Assessment of alternatives to drug deposits |
| Authorized to deposit drugs and pesticides
subject to the conditions in s.5 & s.6 |
| Section 6: Deposit of Pesticides.
1. Complies with Pest Control Products Act
2. Assessment of alternatives to pesticide deposits
|
| Section 7: Measures to minimize detriment from deposit of feces, feed, pesticides and/or drugs. | Required |
| Sections 8 & 9: Baseline monitoring measures required for new (s. 8) or expansion of existing marine finfish sites (s. 9). | Not Applicable | As
required
by provinces | Not Applicable | Required
(unless facility has a standing biomass of 2.5 t or less or an annual production of 5 t or less) |
| Section 10-12: Monitoring of BOD matter deposit | Not required, unless a condition of a provincially issued licence. | Required
(unless facility has a standing biomass of 2.5 t or less or an annual production of 5 t or less) |
| Section 13: Notification of observed morbidity/mortality of wild fish outside the facility | Required
[If drugs or pesticides are not deposited, s.13 does not apply.] |
| Sections 14 & 15: Submission of Annual Report | Required |
| Section 15: Fisheries Act s.35: Prescribed works, undertakings, activities and conditions | Authorized (but does not apply where PAR applies, i.e. in BC)
subject to the conditions under s.15 |
applies to land-based facilities that have the potential to deposit deleterious substances into fish-bearing waters or to cause serious harm to fish that are part of a commercial , recreational or Aboriginal fisheries or the fish that support them.
AAR Application based on type of aquaculture facility
|
AAR Requirements refer to AAR section numbers: | Description | Open-water Shellfish | Freshwater Finfish Open-water facilities | Freshwater Finfish
Land-based facilities | Open-water Marine Finfish Facilities |
| --- | --- | --- | --- | --- | --- |
| Section 5:
Deposit of Drugs | 1. Complies with Food and Drugs Act | 1 | 1 | 1 | 1 |
| Section 5:
Deposit of Drugs | 2. Mitigation measures to minimize accidental deposits | 1 | 1 | 1 | 1 |
| Section 5:
Deposit of Drugs | 3. Assessment of alternatives to drug deposits | 1 | 1 | 1 | 1 |
| Section 6:
Deposit of Pesticides | 1. Complies with Pest Control Products Act | 1 | 1 | 1 | 1 |
| Section 6:
Deposit of Pesticides | 2. Assessment of alternatives to pesticide deposits | 1 | 1 | 1 | 1 |
| Section 7 | Measures to minimize detriment from deposit of feces, feed, pesticides and/or drugs | 2 | 2 | 2 | 2 |
| Section 8 | Baseline monitoring measures required for new marine finfish sites | 3 | 3 | 3 | 6 |
| Section 9 | Baseline monitoring measures required for expansion of existing marine finfish sites | 3 | 3 | 3 | 6 |
| Section 10-12 | Monitoring of BOD matter deposit | 5 | 5 | 5 | 6 |
| Section 13 | Notification of observed morbidity/mortality of wild fish outside the facility | 2 | 2 | 2 | 2 |
| Section 14 & 16 | Submission of Annual Report | 2 | 2 | 2 | 2 |
| Section 15** | Fisheries Act s.35: Prescribed works, undertakings, activities and conditions | 7 | 7 | 7 | 7 |
1. Authorized to deposit drugs and pesticides (subject to the conditions in s.5 & s.6)
2. Required
3. Not applicable
4. As required by provinces
5. Not required, unless a condition of a provincially issued licence
6. Required, unless the facility has a standing biomass of 2.5 t or less or an annual production of 5 t or less
7. Authorized (but does not apply where PAR applies, i.e. BC) subject to the conditions under s.15
applies to land-based facilities that have the potential to deposit deleterious substances into fish-bearing waters or to cause serious harm to fish that are part of a commercial , recreational or Aboriginal fishery or the fish that support* them.
Annex 3: Regional Aquaculture Management Offices
Use this information for the notifications and reports required in AAR sections 6, 8, 9, 12 and 16.
| Region | Contact |
|---|---|
| Central and Arctic Region | |
| Includes Ontario, Manitoba, Saskatchewan, Alberta, Nunavut and Northwest Territories | Regional Aquaculture Management Contact |
| 501 University Cr. | |
| Winnipeg, MB, R3T 2N6 | |
| Tel: (204) 983-5000 | |
| Email: DFO.CAAAR-RAACA.MPO@dfo-mpo.gc.ca | |
| Gulf Region | |
| Extends from the New Brunswick-Quebec border to the northern tip of Cape Breton, and includes the province of Prince Edward Island and parts of New Brunswick and Nova Scotia | Regional Aquaculture Management Contact |
| 343 Ave Université | |
| Moncton, NB, E1C 9B6 | |
| Tel: (506) 874-0362 | |
| Email: DFO.GLFAAR-RAAGLF.MPO@dfo-mpo.gc.ca | |
| Maritimes Region | |
| Extends from the north-eastern tip of Cape Breton to the New Brunswick-Maine border and includes part of Nova Scotia and part of New Brunswick | Regional Aquaculture Management Contact |
| 1 Challenger Drive, 6th Floor, Polaris Building, P.O. Box 1006 | |
| Dartmouth, NS, B2Y 4A2 | |
| Tel: (902) 402-0298 | |
| Email: DFO.MARAAR-RAAMAR.MPO@dfo-mpo.gc.ca | |
| Newfoundland and Labrador Region | |
| Includes the province of Newfoundland and Labrador | Regional Aquaculture Management Contact |
| Northwest Atlantic Fisheries Centre | |
| East White Hills Road, P.O. Box 5667 | |
| St. John’s, NL, A1C 5X1 | |
| Tel: (709) 772-6674 | |
| Email: DFO.NLAAR-RAANL.MPO@dfo-mpo.gc.ca | |
| Pacific Region | |
| Includes British Columbia and Yukon | Regional Aquaculture Management Contact |
| 200-401 Burrard Street | |
| Vancouver, BC, V6C 3S4 | |
| Tel: (604) 666-3152 | |
| Email: DFO.PACAAR-RAAPAC.MPO@dfo-mpo.gc.ca | |
| Quebec Region | |
| Includes the province of Quebec | Regional Aquaculture Management Contact |
| 104 Dalhousie Street | |
| Québec City, QC, G1K 7Y7 | |
| Tel: (418) 648-3236 | |
| Email: DFO.QCAAR-RAAQC.MPO@dfo-mpo.gc.ca | |
| ## Annex 4: Reporting a morbidity/mortality event | |
| Use these numbers ONLY to report a morbidity/mortality event under AAR section 13. | |
| Province/Territory | 1-800 Number |
| --- | --- |
| Alberta | 1-800-265-0237 |
| British Columbia | 1-800-465-4336 |
| Manitoba | 1-800-265-0237 |
| New Brunswick | 1-800-565-1633 |
| Newfoundland and Labrador | 1-800-563-9089 |
| Northwest Territories | 1-800-265-0237 |
| Nova Scotia | 1-800-565-1633 |
| Nunavut | 1-800-265-0237 |
| Ontario | 1-800-265-0237 |
| Prince Edward Island | 1-800-565-1633 |
| Quebec | 1-800-363-4735 |
| Saskatchewan | 1-800-265-0237 |
| Yukon | 1-800-465-4336 |
| ## Annex 5: Drugs and Pesticides Requiring Testing under the AAR (s.13) | |
| Compound | Commercial Name |
| Pesticide (bath) | |
| --- | --- |
| Emamectin benzoate a | SLICE® |
| Teflubenzuron a | Calicide® |
| Ivermectin | Ivomec® |
| Deltamethrin | Alphamax® |
| Azamethiphos b | Salmosan® |
| Cypermethrin | EXCIS® |
| Dichlorovos | Aquaguard® |
| aTeflubenzuron (i.e., Calicide ®) and emamectin benzoate (i.e., SLICE ®) are registered for use in fish in Canada. | |
| bAzamethiphos (i.e., Salmosan®) and hydrogen peroxide-based products (e.g., Paramove 50®) are the only pesticide compounds currently authorized for sea lice treatments in Canada. | |
| Samples collected following Annex 6: Sampling Guidance for Drugs and Pest Control Products (i.e. fish tissue, marine water, and marine sediment) may be analyzed for the compounds listed above as well as other drugs and pesticides as required. | |
| When testing is required, the owner or operator will be provided a list of accredited labs via a Fisheries Act section 36(6). | |
| ## Annex 6: Sampling Guidance for Drugs and Pest Control Products | |
| Note: The following information is offered as guidance and may change, depending on site-specific direction from Fisheries and Oceans Canada. | |
| ### 1. General Methods | |
| 1. All water, sediment, and/or tissue samples collected by the owner or operator will be submitted to a laboratory accredited by the Standards Council of Canada. The specific drugs and pest control products to be analyzed will be determined by DFO (see Annex 5 for a list of drugs and pesticides that may require testing). | |
| 2. Immediately prior to any treatment, prepare all required sampling supplies and equipment as described below and transfer to the collection boat, which should be ready for a rapid deployment. | |
| 3. Copies of the field note forms and specific requirements for chemical analysis should be included with the shipment to the analytical lab. | |
| 4. Original field note forms and digital images should be stored in a secure cabinet. | |
| 5. Samples, sampling supplies, and equipment must be stored in a clean environment, away from drugs and pest control products, to reduce the potential for contamination. Sealed, watertight containers are preferred for storing sampling supplies to help to prevent contamination. | |
| 6. All equipment must be cleaned with detergents and rinsed three times with sterile water between sampling sessions and when there may have been direct exposure to drugs and pest control products or indirectly via dust or water vapour |
2. Fish Samples
- Upon observation of moribund or dead fish of any life stage, and subsequently notifying a fishery officer and obtaining DFO authorization, site staff should immediately deploy the collection boat to locate the fish and begin sampling.
- Fish of the same species and of representative size should be collected, up to a maximum of 50 fish per species. Where permissible, reference fish of similar size and species should be collected in the same manner and numbers from outside the zone of aquaculture influence.
- Protect the integrity of the fish by ensuring no damage occurs to the outside or to the gills of the fish. External lacerations provide entry points for contamination and can lead to fluid loss and/or uncertainties regarding analysis results.
- Using sterile impermeable (e.g. latex or nitrile) gloves set the fish on a clean piece of aluminum foil to record observations.
- Record digital images of all fish, ensuing that any morphological abnormalities and parasites are observed and the corresponding sample identification label is included in each image.
- Double wrap individual fish in heavy duty aluminum foil then double bag the fish using appropriately-sized sterile bags, ensuring that the sample identification label is included inside the outer bag and the bags are sealed with a zap strap. Place clean cork stoppers over significant carapace spines prior to wrapping.
- Store fish on ice in a closed cooler during collection.
- Continue collecting dead or moribund fish observed in the vicinity of the aquaculture site using new gloves, new aluminum foil and a clean club between fish.
- Low temperature storage is important to preserving the target analytes by preventing their degradation. Fish should be kept in bags on ice during collection and subsequently frozen as soon as possible on-site at -20°C for storage.
- As soon as the fish are cooled to -20°C, and within two weeks after collection, fish should be packed in coolers containing dry ice, ice or cooler packs and shipped to a lab for tissue processing, extraction, and analysis, as described below.
- During transport to the lab, frozen fish should be packaged in coolers with ample supply of coolant material. The amount of coolant material required should reflect the sample weight and anticipated shipping time.
- If using dry ice, follow shipping company guidelines.
3. Sediment Samples
- At the same time as the collection of fish samples, sediment samples will be collected.
- Three sediment samples (replicates) for analysis must be collected at the same sampling location(s) as identified for BOD sampling. An equivalent number of reference samples will be collected from an area beyond the predicted exposure zone.
- All equipment parts that come in contact with a sample must be composed of metal, glass, or uncolored polyfluorocarbon.
- Follow the procedures for collecting sediment grabs or cores as per the respective sections of the Monitoring Standard (Annex 7) regarding BOD sampling.
- Remove the top 2 cm of sediment from the core tube.
- Place the sediment into 500 mL amber glass jars. Immediately chill samples by placing them in an ice chest with wet or dry ice for transport, and ship to an accredited Standards Council of Canada laboratory for analysis.
- Sediment samples should be kept on ice and sent to an accredited laboratory for analysis within three (3) days after collection.
4. Water Samples
- Water column sampling at all stations must be completed at the same time as fish and sediment samples.
- If a tarpaulin application method was employed, three water column samples (replicates) will be taken:
- at a horizontal distance of 0 m (i.e., within the cage); and;
- at a horizontal distance 100 m from the cage edge, in the direction of the dominant current, and within 10 m of the surface ; OR
- If a well boat application method was employed, three water column samples will be taken:
- at a horizontal distance of 10 m (i.e., within the treatment well); and;
- at a horizontal distance 100 m from the discharge point, in the direction of the discharge plume; and within 10 m of the surface.
- An equivalent number of reference samples will be collected at the same depth as those collected at the 100 m station.
- Clean-sampling techniques are required when collecting samples for analysis of trace organic compounds and major inorganic elements, particularly when the target analyte may be subject to contamination from field and/or laboratory procedures at a level that could exceed data quality requirements.
- Select equipment with metal, glass, or colourless fluorocarbon polymer components if the components will directly contact samples to be analyzed for organic compounds. Do not use plastics other than colourless fluorocarbon polymers.
- A Van Dorn Sampler is recommended to obtain discrete, surface samples, but any non-contaminating sampler may be used.
- Water samples should be sent to an accredited laboratory for analysis within 24 hours of collection. The protocol for collection, preservation, and transport should be reported in the AAR Annual Report to DFO.
5. Records
- For the proper interpretation of analytical results it is important to collect all vital field data in a consistent and standardized method.
- Data collection forms should include the specific information included below. Separate identification labels that accompany the samples during storage and shipment should be designed to correspond with the data collection forms.
- Indelible pens and field grade waterproof paper are recommended.
6. Specific information requirements include:
- Aquaculture site and company name;
- Time and location when fish were first observed and name of observer(s);
- Contact information;
- Depth and GPS coordinates of aquaculture site;
- Species, size and biomass of fish on site at the time of treatment;
- The drug or pest control product , method of application, application concentration, amount of product applied and stage of application when affected fish were observed;
- Date, time and location (GPS coordinates) of sample collection and names of collectors;
- Surface water and air temperature at time of sampling;
- Weather observations
- Collection method;
- Sample Identification Number for each fish;
- Photograph number;
- Length (cm);
- Weight (g);
- Species collected;
- Condition: live or dead and, if live, observed behaviour;
- Observed morphological abnormalities, such as evidence of tissue damage, growth abnormalities, lacerations, fin erosion, skin ulcers, neoplasms;
- Quantity and type of ectoparasites; and
- Record of storage and transport conditions (time spent at ambient temp/time and temperature spent on ice at collection, and storage time).
Annex 7: Monitoring Standard
2018 Aquaculture Monitoring Standard (PDF 138 KB)
Introduction
This Aquaculture Monitoring Standard (monitoring standard) will support the monitoring and sampling requirements of the Aquaculture Activities Regulations (AAR) under the Fisheries Act. This document provides the necessary level of detail to enable the owner or operator of an aquacul-ture facility to produce to the greatest extent possible, consistent and high quality data to support the implementation of the AAR. This monitoring standard will be amended (in consultation with provinces) from time to time.
Definitions
Containment array means an assembly of interconnected cages used to cultivate fish.
Marine worms mean aggregates of opportunistic polychaetes that colonize organically enriched substrates
Reference station means a sampling station chosen to represent a background or natural state. Reference stations may be chosen as discrete sampling locations or contained along a transect representing a traditional “gradient” to “background” approach.
Sampling station means the location where recording is carried out and any samples are collected.
Soft bottom site means, further to definition in AAR, a site where acceptable samples can be taken from the benthic substrate based on the grab acceptability criteria and sampler options re-lated to oceanographic conditions and substrate type or classified as soft bottom based on pro-vincial criteria.
Transect means a directional line, along which sampling stations are established or visual observations are made.
I. Survey for baseline information for new sites and expansion of existing sites [AAR section 8 and 9]
Predicted contours [AAR paragraph 8(1)(a)]
- 1. (1) Calculate the rate of deposition of biochemical oxygen demanding (BOD) matter from the facility during maximum daily quantity of feed usage, using an aquaculture waste deposition model, and map the 1, 5, and 10 grams carbon per meter squared per day (g C/m2/day) depositional contours.
- (2) Site-specific oceanographic data will be used for model inputs. Using an aquaculture waste deposition model, input characteristics for food and fecal waste must be either accepted international standard values or operator measurements. Model simulations are not to include estimates of post-deposition particle resuspension.
Survey of fish and fish habitat [AAR paragraph 8(1)(b)]
- 2.(1) When conducting surveys of fish and fish habitat, sampling stations along a transect or within a grid must be representative of the different physical/biological characteristics of the bottom habitat within the modeled 1 g C/m2/day depositional contour and the entire lease, including reference stations where applicable.
- (2) Sampling is not required within areas of the tenure or 1 g C/m2/day depositional contour where depths exceed 300 m.
- (3) If historical fish and fish habitat data for an expanded lease area demonstrates a homogeneous coverage of mud or sandy habitat, then only one video transect across the width of the entire lease in the direction of the dominant current and under the proposed array location is required.
- (4) The fish and fish habitat benthic survey is to be conducted to identify species that are 1 cm or greater in length and the type of habitat present.
- (5) All fish habitats/substrates must be identified.
Bathymetry survey [AAR paragraph 8(1)(c)]
- 3.(1) A bathymetric survey must be conducted with a minimum resolution of 10 m contours to generate depth profiles, related to chart datum, within the modeled 1 g C/m2/day depositional contour, as calculated in subsection 1(1), and covering the entire lease, including reference stations where applicable.
- (2) Despite subsection 3(1), a bathymetric chart from the Canadian Hydrographic Service that includes depth profile contours in 10 m increments may be used instead of conducting a bathymetric survey.
- (3) Echosounders, transducers, and associated equipment used to generate the bathymetric survey in subsection 3(1) must be calibrated to industry standards.
- (4) Horizontal position fixing measurements must be carried out using a differential Global Positioning System (dGPS).
Benthic substrate monitoring [AAR paragraph 8(1)(d)]
- 4.(1) Information concerning the seabed will be collected within locations representative of the entire lease and the modeled 1 g C/m2/day depositional contour, as calculated in subsection 1(1), including reference stations where applicable.
- (2) In the case of an aquaculture facility that is located in tidal waters in or adjacent to:
- Quebec, Nova Scotia, New Brunswick, Prince Edward Island or Newfoundland and Labrador, collect samples of the benthic substrate in the center and at each corner of the lease boundary
- New Brunswick, collect samples of the benthic substrate at the end of a 50 m transect from the lease boundary, in the direction of the dominant current
- Quebec, Nova Scotia, or Prince Edward Island, collect samples of the benthic substrate between 100 to 300 m from the edge of the lease boundary, in the direction of the dominant current
- British Columbia, collect samples of the benthic substrate at a minimum of two sampling stations (30 m and 125 m away from the cage edge) along two transects that align with the area of greatest predicted impact and with the dominant and sub-dominant current directions
- If the containment array mentioned in paragraph 4(2)(d) is greater than 200 m in length and its long axis is perpendicular to the direction of the dominant current, additional sampling is required in the following manner:
- for every 200 m increment in length, establish additional transects with sampling stations at 30 and 125 m adjacent to each transect established in paragraph 4(2)(d)
- transects must be parallel to each other and a minimum of 50 m apart
- based on the 5 g C/m2/day contour, as calculated in subsection 1(1), the transects are to be located in the direction of maximum deposition as determined by the 5 g C/m2/day contour
- (3) In addition to the criteria specified in the AAR subsection 10(2), the following relevant criteria must be complied with for benthic substrate sampling using grab or core devices:
- Obtain dGPS coordinates at each corner of the containment structure array and at all sampling stations, Readings are to be recorded in decimal degrees or Universal Transverse Mercator coordinates, using the North American Datum of 1983 as reference.
- The sampling device shall be of a weight and size to ensure sampler decent is vertical and directly below the area of deployment with no evidence of drift (angle in line is not noted).
- Collect at least 3 samples from each station. At least 5 failed attempts per station must occur and be documented before sediment sampling is abandoned.
- Overlying water is present (indicates minimal leakage) and must be removed prior to processing and storage by siphoning, not decanting.
- The overlying water is clear or not excessively turbid.
- The sediment-water interface is intact and relatively flat, with no sign of channeling or sample washout.
- There is minimal sediment loss.
- Obtain successive samples from substrate that has not been disturbed by previous sampling.
- Dispose of excess sediment in a manner that minimizes the possibility of contaminating subsequent samples.
- Collect at least one subsample that is representative of the top 2 cm of sediment within the sampler.
- Remove all non-sedimentary material, including all large shell fragments, fish, wood waste, and rock, before placing material in containers for analyses.
- Keep sampler level when retaining samples.
- Remove overlying water quickly from sample and avoid areas for subsampling where any overlying water may remain.
- The sampler is not overfilled so that the sediment surface is touching the top of the sampler.
- The core sampler was not inserted at an angle or tilted upon retrieval.
- All sampler attempts and sediment collections must be clearly documented by video or still pictures to support sampling approach acceptability criteria.
- (4) The following information concerning the seabed must be collected with samples collected:
- latitude/longitude using dGPS
- depth
- date and time of sampling
- sediment texture and colour
- photo of sediment sample
- presence of gas bubbles
- estimation of surface coverage of bacterial mats
- estimation of surface coverage marine worms
- presence of fish feces and feed
- presence of flocculent organic material
- free sulfide
- redox
- sediment grain size; and
- percent organic matter and porosity only in Quebec, Nova Scotia, New Brunswick, Prince Edward Island, and Newfoundland and Labrador; or
- total volatile solids in British Columbia
- (5) The following procedures must be complied with when measuring the concentration of free sulfide, redox, and sediment grain size in sediment samples:
- The concentration of free sulfide in the sediment samples must be determined within 36 hours of collection, except in British Columbia where sediment samples must be analyzed within 5 minutes.
- If the free sulfide concentration measurements are not carried out within 5 minutes of the sample being collected, subsamples must be stored between 2 and 5 °C until they are analyzed.
- Use a silver/sulfide probe with a sulfide sensitivity range of at least 0 to 19,900 μM and a accuracy of ± 5 % with an appropriate ion-selective electrode (ISE) or millivolt (mV) meter of 0.1 mV resolution.
- The probe must be calibrated using three to five serial dilutions of a standard sulfide solution, as required, beginning with the most dilute solution.
- Sediment grain size distribution measurements must be recorded according to the Wentworth grain size scale as a percentage of the total subsample mass.
- Redox measurements must be conducted in the following manner:
- The redox combination electrode must have an appropriate ISE or mV meter.
- Calibration of the probe must be completed before measurements are taken.
- Redox values and temperature must be measured at the same time as the free sulfide.
- (6) Where samples cannot be obtained as specified in subsections 4(2) and (3), visual monitoring must be conducted as specified in sections 5 and 6.
Exception - (7) Sampling is not required within areas of the tenure or 1 g C/m2/day depositional contour where depths exceed 300 m.
Visual monitoring procedures [AAR paragraph 11(2)(a), (b), and (c)]
- 5. (1) Visual monitoring of the benthic substrate must be conducted in the following manner:
- Must use a handheld or dropped still camera, or diver-operated, towed, or remotely operated video camera.
- The weight of towed devices must allow for stable movement at a consistent depth without disturbing sediment substrate.
- Cameras used for underwater photography must illuminate the benthic substrate to an intensity balanced to the optical sensitivity of the lens such that a uniform field of view is visible.
- The optical resolution of the camera lens must be sufficient that organisms greater than 1 cm in size can be distinguished by the unaided eye and identified in a 0.5 m x 0.5 m horizontal field of view; magnification of species observed is acceptable, as necessary, for identification purposes.
- Diver-operated, towed, or remotely operated video cameras must be operated at a constant velocity that permits accurate identification of visual parameters.
- Original video must be transferred to digital format storage media with no post-survey video editing.
Visual monitoring locations [AAR paragraph 11(2)(a)(b) and (c)]
- 6. (1) In the case of an aquaculture facility that is located in tidal waters in or adjacent to Quebec, Nova Scotia, New Brunswick, or Prince Edward Island, images will be taken at the same stations specified in paragraph 4(2)(a).
- (2) In the case of an aquaculture facility that is located in tidal waters in or adjacent to Newfoundland and Labrador, images must be recorded based on a 100 m2 grids within the lease. At least one station must be established at the edge of the proposed containment array (0 m).
- (3) In the case of an aquaculture facility that is located in tidal waters in or adjacent to British Columbia, images must be recorded along two transects that start at the edge of the proposed containment array, align with the area of greatest predicted impact and with the dominant and sub-dominant current directions and extend for a minimum of 140 m, with a maximum deviation of ± 20% from that bearing. Images must also be collected from reference stations during baseline surveys.
- (4) Coordinates as established by a dGPS must be noted at the start and finish of each transect. The readings must be recorded in degrees minutes decimal minutes with 3 digits following the decimal point, using the North American Datum of 1983 as reference.
Exception - (5) Sampling is not required within areas of the tenure or 1 g C/m2/day depositional contour where depths exceed 300 m.
Timing of sampling [AAR paragraph 8]
- 7. Benthic substrate surveys or visual monitoring must be conducted prior to the introduction of fish to the site.
Recording of baseline survey information [AAR subsections 8(1) and (3), and 9(1)]
- 8. The report of the baseline survey findings must include at a minimum:
- The observations recorded from the underwater surveys, including the unedited recorded images
- A map with the locations of transects and habitat information generated during these transects at a minimum resolution of 1:5000, the bathymetry of the seabed at a resolution of 10 m contours; and the estimated footprint of deposition of biochemical oxygen demanding matter in 1, 5, and 10 g C/m2/day contours
- The results of sediment sampling as specified in subsection 4(4) or records of failed sediment sampling attempts as per 3(c)
- Unedited visual recordings must be submitted with the following information:
- positional data including all start and stop points and way points in between using corrected dGPS
- ocean depth of the location recorded; and for continuous video depths at a minimum of 10 m intervals
- date and time of sampling
- estimation of surface coverage of bacterial mats
- estimation of surface coverage of marine worms
II. Procedures for operational monitoring [AAR section 10(1)]
Timing of sampling [AAR paragraphs 10(1)(a) and (b)]
- 9. Benthic monitoring samples or video must be taken at the facility at least once during the production cycle at sea or every 24 months for farms with finfish continuously on site:
- (1) in British Columbia, within 30 days of peak feeding or peak biomass
- (2) in Quebec, Nova Scotia, New Brunswick, Prince Edward Island, or Newfoundland and Labrador, between July 1 and October 31, close to peak feeding
- (3) during times when water and weather conditions minimize adverse effects on sampling quality and facilitate accurate sampling at stations
Benthic substrate monitoring [AAR paragraphs 10(1)(a) and (b)]
- 10. (1) Where benthic substrate monitoring is conducted, the procedures as specified in section 4, excepting paragraphs 4(2)(a) and (b), must be followed. Sampling is not required within areas of the tenure or 1 g C/m2/day depositional contour where depths exceed 300 m.
- (2) Specifically for Quebec, Nova Scotia, New Brunswick, or Prince Edward Island, Table 1 is used to determine the number of sediment samples at 0 m stations and transects.
Exception - (3) Sediment grain size is not required to be measured during operational monitoring.
Visual monitoring [AAR paragraph 11(2) (a)(b) and (c)]
- 11. (1) The procedures in subsection 5 will support compliance with the facility restocking threshold.
- (2) In the case of an aquaculture facility that is located in tidal waters in or adjacent to Nova Scotia or New Brunswick:
- Observations from each transect will be made at 0 m, 10 m, 20 m, 30 m, 40 m and 50 m locations
- The number of transects are indicated in Table 1, Column II based on the number of fish stocked, specified in Table 1, Column I
- Cages along the outside perimeter of the cage configuration must be those selected for positioning of transects. Transects will be positioned starting with the cage with the highest biomass and proceeding in descending order, and in the direction of the prevailing water current
- Compliance stations include all stations between 0 and 50 m
- (3) In the case of an aquaculture facility that is located in tidal waters in or adjacent to Newfoundland and Labrador:
- Observations must be recorded along a minimum of 6 transects, consisting of two perpendicular transects from each corner of the containment array and extending away from the array for 100 m with 20 m increments
- Observations from each transect will be made at 0 m, 20 m, 40 m, 60 m, 80 m and 100 m locations
- Additional transects must be recorded from and perpendicular to the middle of each side of an array that consists of more than 9 cages in a row
- Compliance stations include all stations between 0 and 100 m
- (4) In the case of an aquaculture facility that is located in tidal waters in or adjacent to British Columbia, follow subsection 6(3) for sampling locations.
Exception - (5) Sampling is not required within areas of the tenure or 1 g C/m2/day depositional contour where depths exceed 300 m.
Reference stations [AAR subsections 10and 11]
- 12. (1) At least one reference station must be established as required according to the following criteria in the case of an aquaculture facility that is located in tidal waters in or adjacent to British Columbia, Quebec, Nova Scotia, New Brunswick, Newfoundland and Labrador, or Prince Edward Island:
- Either consisting of a transect outward from the lease boundary or as discrete stations that are not exposed to biochemical oxygen demanding matter deposited from the facility
- In British Columbia, these are discrete stations between 0.5-2.0 km from the facility location
- The reference station depth is within ± 10 m of the range of depths of the sampling stations as specified in subsection 4(2) and section 6
- Topography, seabed type, current and tidal regimes, sediment grain size, and the amount of freshwater runoff influence are to be representative of the sampling stations, and
- If it is determined that the seabed is composed of predominantly soft sediment, a minimum of one reference station will be established with a minimum of 3 sediment samples collected per station
- If the seabed is determined to be predominantly not soft sediment, visual monitoring must be conducted as specified in section 11
Recording of operational survey information [AAR subsection 16]
- 13.(1) Recording of benthic surveys as per section 8.
Exception - (2) BOD modelling, fish and fish habitat surveys and bathymetric mapping is not required for operational monitoring.
Table 1: Number of sampling stations required for sediment and visual sampling (Nova Scotia and New Brunswick)
| Column I | Column II | Column III | Column IV |
| --- | --- | --- | --- |
| Maximum number of fish within cage site array during production cycle | Number of transects | Number of sampling stations (not including reference stations) | Number of samples (3 samples/station for soft bottom sites at 0 m from containment array) |
| 1-200,000 | 2 | 2 | 6 |
| 200,001-300,000 | 3 | 3 | 9 |
| 300,001-400,000 | 4 | 4 | 12 |
| 400,001-500,000 | 4 | 5 | 15 |
| 500,001-600,000 | 4 | 6 | 18 |
| 600,001-700,000 | 4 | 7 | 21 |
| 700,001-800,000 | 4 | 8 | 24 |
| 800,001-900,000 | 4 | 9 | 27 |
| 900,000-1,000,000 | 4 | 10 | 30 |
Annex 8: Program Protocols for Marine Finfish Environmental Monitoring in British Columbia
Acronyms, Abbreviations and Definitions
ANOVA: analysis of variance
Beggiatoa-like species: species of bacteria that form visible mats on the seabed surface in areas of organic enrichment which may be in the genus Beggiatoa, but may also include those found in different genera.
Benthic Monitoring: sediment sampling and video surveys conducted during operation of a finfish aquaculture facility. This includes operational monitoring as well as prescribed follow-up and pre-stock monitoring.
dGPS: differential Global Positioning System
DI: de-ionized
EDTA: Ethylenediaminetetraacetic Acid Disodium Salt Dihydrate
Eh: redox potential (expressed in millivolts, mV)
Epifauna: animals that live on top of the substratum
HA: alternate hypothesis
HO: null hypothesis
High slack tide: is that point in time in any given location where the water depth has reached its maximum height (above chart datum) and any water movement has ceased, up until the current reverses direction. Official tide tables are to be used to determine the predicted time for maximum height of the nearest tidal station and then extrapolate for the specific aquaculture facility location.
Infauna: animals that live within the substratum
M: median
μM: micromolar; unit of measure used to express sulfide concentrations
Macrofauna: animals with body sizes on the scale of mm
Megafauna: animals with body sizes on the scale of cm
n: sample size
NAD: North American Datum
OPC: Opportunistic Polychaete Complexes
Post Processed (PP): GPS positions are corrected for satellite and propagation errors after the observations are made.
QA/QC: Quality Assurance / Quality Control
Real Time (RT): GPS positions are corrected for satellite and propagation errors as the observations are made. The reference receiver sends corrections and/ or observations to the roving receiver.
ROV: Remotely Operated Vehicle
S=: free sulfide concentration (expressed in micromolar; μM)
SAOB: Sulfide Anti-Oxidant Buffer
SD: Standard Deviation
SGS: Sediment Grain Size
TVS: Total Volatile Solids (expressed as a percentage)
x: sample mean
1. Compliance Sampling Locations
1.1 Selecting compliance sampling stations or zones:
* (a) Use a differential Global Positioning System (dGPS) to obtain readings from each corner of the containment structure array at high slack tide, and ensure this is consistent with the approved location of the array. Note that only corrected dGPS coordinates will be accepted, unless otherwise directed by the Department;
* (b) Use electronic mapping procedures to establish the compliance sampling stations or zones as required for soft or hard ocean bottom substrate sites;
* (c) Locate the compliance sampling stations or zones in areas where the greatest concentration of farm derived organic material is expected taking into consideration the following factors:
* (i) most recent peak biomass sampling and monitoring data;
* (ii) historic sampling and monitoring data;
* (iii) company internal in-cycle monitoring data;
* (iv) DFO audit data;
* (v) modeling data;
* (vi) observed localized current direction; and
* (vii) other direction by the Department;
* (d) Locate the compliance sampling stations or zones along transects in the directions of the dominant and subdominant currents, unless otherwise directed by the Department. A minimum of two transects per containment structure array must be sampled;
* (e) If the containment structure array is perpendicular to the dominant current direction and is greater than 200 m in length, the following is required:
* (i) an additional two transects for every 200 m increment;
* (ii) transects must be a minimum of 50 m apart; and
* (iii) the location of the secondary transects shall be based on modeling or other assessment
methods indicating the location of the 5 g C/m2/day contour as follows:
- if the 5 g C/m2/day contour extends beyond 30 m from the containment structure array on the shoreward end of the array, the transects shall be located such that they transit the contour at its furthest extent; and
- if the 5 g C/m2/day contour does not extend beyond 30 m from the containment structure array on the shoreward end, the transects shall be located on the seaward end of the array;
* (f) If adjacent containment structures or containment structure arrays are less than 60 m apart they will be considered to be a single array when transects are located; and
* (g) Compliance stations or zones are to be identified as follows:
* (i) transects located along the dominant current direction shall be labelled A and C; and
* (ii) transects located along the subdominant current direction shall be labelled B and D; or
* (iii) as otherwise directed by the Department.
1.2 Selecting Reference Stations
* (a) Locate stations 0.5 – 2.0 km from the nearest containment structure associated with the facility;
* (b) Reference stations must be a minimum of 0.5 km apart;
* (c) Ensure mean depth is within 25% of the mean depth of all facility stations. This may require additional reference stations if there is significant variability in the depth associated with the facility stations;
* (d) Ensure that characteristics such as topography, seabed type, current and tidal regimes, amount of freshwater run-off and other applicable characteristics are similar to those at facility stations;
* (e) Ensure that the reference stations have similar effects if facility stations appear to have been influenced by anthropogenic activity (e.g. log dumps); and
* (f) For soft ocean bottom substrate sites, ensure the silt/clay fraction of Sediment Grain Size (SGS) is within 15% of the facility station’s silt/clay fraction. This may require additional reference stations if there is significant variability in the silt/clay fractions associated with the facility stations.
Note : For baseline surveys, reference stations require both sediment analysis and at least 100 m of video data collection. For more information, see application guidance document.
2. Benthic Monitoring and Reporting Requirements
2.1 Monitoring Frequency and Timing * (a) “Peak biomass” monitoring must be conducted: * (i) within 30 days either side of peak biomass for farms that have a production cycle ending in the complete removal of all fish ; or * (ii) at the end of every 24 month period if the production cycle is longer than 24 months; or * (iii) every 24 months for brood stock farms and/or with finfish continuously on site. * (b) Prior to fish entry, pre-stock monitoring must also be conducted as directed by the Department.
2.2 Monitoring Method * (a) Collect sediment samples following the procedures outlined in Section 4. Sediment Sampling except when: * (i) Acceptable grab samples as described below in Section 4.4 (c, d, and e) cannot be obtained at the 0 or 30 m station(s). In this case, report the latitude/longitudes of the locations of the attempted grabs and submit a photo(s) of any sediment within the grab at the failed sampling location(s) to confirm the hard bottom nature of the station(s); or * (ii) Acceptable grab samples as described below in Section 4.4 (c, d, and cannot be obtained at the 125 m station(s). In this case, report the number and locations of the attempted grabs and conduct video surveys following the protocol outlined in Section 3. Video Surveys; or * (iii) Any station is confirmed by the Department as having hard ocean bottom substrate. In this case, grabs do not need to be attempted at that station in the future.
2.3 Report Frequency and Timing * (a) Benthic monitoring reports shall be submitted: * (i) within 30 days of sediment sampling for soft ocean substrate sites; * (ii) within 60 days of video surveys for hard ocean substrate and mixed ocean substrate sites. * (b) Biological monitoring reports shall be submitted: * (i) within 6 months of collecting samples submitted for taxonomic identification; and * (ii) within 14 days of receipt of the results from the taxonomist. * (c) All of the above reports shall also be submitted a minimum of 14 days prior to planned fish re-entry on site.
2.4 Report Content and Format * (a) Reports must be submitted in a format acceptable to the Department and include completed data templates where provided by the Department; * (b) Maps at a scale of 1:5000 must be submitted showing the number and location of containment structures and indicating which pens were stocked during the production cycle. Any harvest pens or transfer pens on-site should be included; * (c) Maps must show the start and endpoints of both sections of each video transect and their associated dGPS coordinates, all compliance sampling stations, the corners of the containment structure array, the associated anchor line locations, dominant current direction, and 10m depth contours; * (d) Peak biomass (in tonnes) must be reported; * (e) For hard ocean bottom substrate sites, high resolution DVDs are to be submitted with video data and the survey data associated with the classification database; * (f) For soft ocean bottom substrate sites, all raw data recorded during sampling and associated chemical analysis must be submitted in the sediment chemistry data template provided by the Department; and * (g) A written summary report, including any required statistical analysis, must also be submitted for all benthic monitoring.
3. Video Surveys
The focus of these protocols is to describe the accepted method of performing continuous video surveys using a remotely operated vehicle (ROV). Standard deployment and positional methods as well as equipment specifications are detailed. 3.1 Monitoring Design * (a) Each transect must start at the edge of the containment structure and extend: * (i) 140 m from the containment structure array; or * (ii) 24 m or six time segments (see Section 3.7 (c)) beyond where Beggiatoa -like species or OPC was last observed to exceed 10% cover, whichever is the greatest distance. * (b) Each transect will consist of two sections described as follows: * (i) 0 – 80 m; an * (ii) 80 m – to transect termination point as described in (a) above.
3.2 ROV Minimum Requirements * (a) Horizontal and vertical thrusters. A lateral thruster is recommended; * (b) A minimum of biannual calibration of: * (i) ROV compass to the accuracy of +3°. Surveys shall be recorded in accordance to the calibrated bearing. * (ii) depth gauge accurate to +2% full scale calibration using a minimum of three depths between 10 m and 100 m. * (c) Paired auxiliary lights: * (i) positioned laterally and on the same plane as camera to provide uniform illumination of a minimum 0.5 m in width for at least 75% of the field of view; and * (ii) illumination intensity is to be balanced with the light sensitivity of camera; and * (iii) light diffusers used to reduce backscatter and shadowing as necessary; * (d) Paired laser lights: * (i) should be used to provide a horizontal scale and estimate of the field of view; and * (ii) the distance between lights should be recorded as it can differ between units.
3.3 Camera and Recording Minimum Requirements * (a) Resolution and illumination of the video quality must be such that biota 1-2 cm in size may be distinguished and identified within a 0.5 m horizontal field of view; * (b) Real time video display with time, depth and compass bearing; * (c) Transect should be identified; and * (d) Original video must be transferable to digital-format storage media (i.e. no post survey video compression).
3.4 Positioning Equipment Requirements * (a) A GPS field unit that facilitates both real-time (RT) differential corrections and post-processed (PP) differential corrections; * (b) GPS positioning software capable of PP differential corrections of the field unit data. If this is not available, report alternate positional methodology and identify whenever uncorrected positions are used; and * (c) An acoustic positioning system for mapping the ROV transect is recommended. If an acoustic positioning system is not available, marker buoys may be used to navigate to transect section start points.
3.5 Conditions and Requirements * (a) Operate ROV during slack tide and/or minimal current conditions to minimize drifting; * (b) Video transects are to begin at edge of containment structure array; * (c) Each transect is to be comprised of two sections: 0 – 80 m and 80 – 140 m (minimum); * (d) If required for clarity, observed compliance factors such as Beggiatoa-like species, OPC, fish feed or feces should be magnified to facilitate identification; * (e) Optimum ROV speed over seabed is 0.2 m/sec. The acceptable range of speed is between 0.15 – 0.25 m/sec; and * (f) Maximum variance from planned transect is +/- 10% of planned transect length and +/- 20% of planned transect bearing.
3.6 Deployment * (a) Deploy weighted line with marker buoy adjacent to containment structure array at point where transect is to start; * (b) Deploy second weighted line with marker buoy on projected transect bearing 80 m from first line using navigational aids such as GPS and navigational software and range finder to confirm distance; * (c) Fly ROV from moored vessel to ocean floor following weighted line adjacent to containment structure array; * (d) Use pre-determined compass bearing to fly ROV toward 80 m weighted line. Estimate distance using time and deployed tether length. Bring ROV to surface and calculate variances for length and width; and * (e) Use predetermined compass bearing fly ROV along transect an additional 60 m (140 m from containment structure array). Bring ROV to surface and calculate variances for length and width. * Note: The length of the second section of transect will depend on compliance with OPC and Beggiatoa-like species standards.
3.7 Video Assessment
* (a) Classification and video assessment must be based on Hard Seabed Aquaculture Video Monitoring Classification Tutorial DVD June 30, 2009 (Revised February, 2011);
* (b) The following information must be included in raw data tables for both peak biomass and prestocking monitoring reports:
* (i) Abiotic parameters
- Substrate using Wentworth scale indicating habitat type
- Location and type of farm litter and debris
* (ii) Biotic parameters
- Percent cover of compliance indicators: Beggiatoa-like species and OPC. All observed Beggiatoa-like species and aggregated polychaetes must be reported.
- Percent cover of fish waste and feces
* (iii) Presence and relative abundance of sensitive, opportunistic and resource/conservation taxa;
* (c) Classify the video imagery into 20 second time segments, assuming speed over ground to be 0.2 m/s unless otherwise specified. A segment length of 20 seconds equates to a linear distance of 4 m; if ROV speed of 0.2 m/sec cannot be maintained, the time of each segment should be adjusted such that each segment represents 4 linear m. A table of stoppages may be included to justify segment length if consistent speed cannot be maintained, or to include ROV ascents/descents. Divide each 20 second segment into 5 second intervals to improve accuracy of the segment classification. Ensure no “overlap” of 20 second video segment classification occurs;
* (d) Calculate length of video transects using the time at start and end points, subtracting all camera stoppages of more than 3 seconds and report temporal length that encompasses the zone of compliance;
* (e) Assess and record only the bottom 2/3 of the screen. The area considered to be “near field”; and
* (f) Input data into the Hard Seabed Aquaculture Classification Database Version 2.2 June 30, 2009 (Revised February, 2011; use the report generating function in the database and the report templates to submit the data.
4. Sediment Sampling
For the purposes of this protocol, sediment sampling is primarily used for compliance purposes. The regression approach is used such that data are collected at stations along transects extending outward from the facility along prevailing current directions. This section describes the protocols on how to choose a sample station, obtain and handle samples, use electrodes and generate physical and chemical data. 4.1 Monitoring Design * (a) The following compliance sampling stations must be sampled along each transect: * (i) At the edge of the containment structure array (0 m station); and * (ii) 30 m from the edge of the containment structure array; and * (iii) 125 m from the edge of the containment structure array. * i. For 30 m and 125 m compliance sampling stations not already predetermined by way points, use range finder to locate stations. (It is recommended that the vessel is tied off to the containment structure array to maintain position). * (b) Two reference stations must be sampled if required by licence conditions; * (c) A minimum of three replicate samples shall be taken at each required compliance sampling station; * (d) The samples reported shall be the first samples that meet quality standards (See Section 5.5); * (e) If the free sulfide concentration in any of the three samples exceeds the threshold for that station, an additional two samples shall be taken and analyzed for free sulfides and redox potential; and * (f) Best survey practices include sampling during slack tide and in low wind conditions to minimize drift and facilitate accurate sampling of predetermined compliance sampling station locations.
4.2 Equipment Requirements * (a) Acceptable soft sediment sampling devices for physical, chemical and biological variables include those where multiple samples can be taken from a single grab: including the Ponar, Smith-MacIntyre, Van Veen; * (b) Meters and electrodes to measure sulfide (‘S’) and redox (Pt). See Section 7 for standardization and calibration procedures; and * (c) An independently powered davit system is recommended.
4.3 Sampling Preparation and Information * (a) Prepare the ‘S’ stock solution (10-2 M Na2S) and SAOB (EDTA/NaOH) solutions in advance (See Section 7); * (b) Add L-Ascorbic acid to the SAOB solution just prior to calibration of silver/sulfide (Ag+/S=) electrode; * (c) If the Ag+/S= electrode requires the filling solution to be topped up, ensure the solution is added at least 30 minutes prior to electrode use to ensure stability of electrode; and * (d) Calibrate Ag+/S= electrode and standardize redox (Eh) electrode just prior to sampling, using protocols outlined in Section 7.
4.4 Sample Collection and Description
* (a) Decrease descent speed when approaching the seafloor and retrieve the sampling device at sufficiently slow speeds to preserve the intact sediment surface structure in the sample;
* (b) Record the following and include in the raw data table template submitted with monitoring reports:
* (i) latitude/longitude of sample locations using corrected dGPS ;
* (ii) Magnetic and true-north bearings of transects (should be based on moving away from farm);
* (iii) ocean depth; and
* (iv) time that sample was successfully obtained;
* (c) If the sample appears compromised by a previous grab (i.e., was collected in a divot formed by previous sampling), discard this sample and try again;
* (d) Ensure excess sediment from grabs is disposed of in a manner such that it will not compromise subsequent grabs;
* (e) Check for indicators of an acceptable grab sample:
* (i) overlying water present – indicating minimal leakage and associated disturbance;
* (ii) overlying water not excessively turbid - indicating minimal sediment disturbance;
* (iii) sediment surface relatively flat – indicating minimal sediment loss due to wash-out; and
* (iv) minimum penetration depth of 5 cm for surficial sediments is achieved;
* (f) If sampling device is overfilled, remove some or all of the detachable weights and/or reduce deployment rate;
* (g) Do not make more than 4 attempts to obtain a suitable sample grab at any sampling station location. If unsuccessful:
* (i) Move vessel over 1 – 3 m and attempt to obtain sample. If unsuccessful after one attempt;
* (ii) Move vessel in opposite direction from original location and attempt to obtain sample;
and
* (iii) When a sediment sample cannot be taken, refer to Section 2.2;
* (h) If samples for macrofauna analysis are to be collected, siphon the overlying water from the sample and retain for sieving;
* (i) Record the following information and include in the raw data table template submitted with the monitoring report:
* (i) depth of sediment in cm;
* (ii) sediment texture;
* (iii) sediment colour and presence of black sediment;
* (iv) odour on a scale of 0 – 4, with 4 being the strongest;
* (v) presence of gas bubbles;
* (vi) presence of Beggiatoa -like species;
* (vii) presence of Opportunistic Polychaete Complexes (OPC);
* (viii) presence of fish feed;
* (ix) presence of fish feces;
* (x) presence of flocculent organic material;
* (xi) macrophytes;
* (xii) megafauna;
* (xiii) terrigenous material;
* (xiv) farm litter; and
* (xv) Take a colour photo of the sample and include it in the monitoring report.
4.5 Preparing Sediment Subsamples Perform Eh and ‘S’ analyses within 5 minutes after sample collection by completing the following steps: * (a) Collect duplicate subsamples from the top 2 cm of sediment from the centre of each side of the grab using a plastic spatula, shallow spoon, or cut-off plastic syringes if sediment texture permits. A minimum volume of 25 mL is required for each Eh and ‘S’ analysis; (b) Place both sub-samples in a suitable container and remove all unrepresentative material (e.g. large shell fragments, megafauna, wood waste, rock). Then homogenize the subsamples by gently mixing the sediment; and * (c) Remove the sediment needed for Eh and ‘S’ analyses. The remaining sediments are to be placed in whirl packs or tissue cups for lab analysis. TVS and percent moisture (water content) analysis can be done on the same sample. SGS require separate subsamples for analysis. All storage containers must be kept cool and be air-tight to prevent desiccation and exposure to excess air.
4.6 Measurement of ‘S’
* (a) In order to measure ‘S’ a 1:1 volume ratio of SAOB to sediment is required;
* (b) Place 10 mL of SAOB in a small plastic or glass graduated container and then add sediment from section 4.6 (c) until the meniscus is at the 20 mL gradation. (Always add SAOB first);
* (c) Briefly stir the mixture and then insert the Ag+/S= electrode into the sample. Ensure the tip of the electrode is fully covered by the SAOB/sediment slurry;
* (d) Gently move the electrode in the slurry until electrode stabilizes. Depending on electrode a “Ready” message appears on the meter screen and/or a beep is heard (typically 1 – 4 minutes).
Note: Solid phase metal-‘S’ complexes may be solubilised under alkaline conditions and prolonged exposure of sediment to SAOB before the electrode potential is recorded should be avoided;
* (e) Gently wipe the electrode and remove all sediment prior to insertion into the next sample;
* (f) Any oily residue on the electrode must be removed prior to further use; and
* (g) If potentials recorded with the Ag+/S= electrode in a sediment sample do not stabilize in 1 – 2 minutes, performance can be assessed without a full recalibration. The ‘S’ concentration can be measured in one of the dilute ‘S’ standards if it has been stored in a dark cooled container with a minimum head space. Do not attempt to correct the data for drift if deviations are less than 10 – 20% of expected standard values. If measured values have a greater deviation than the expected value a full recalibration should be performed following protocols in Section 7.
4.7 Measurement of Eh
* (a) After the subsample for ‘S’ is removed for the analysis in Section 4.6, place the redox electrode in the remaining subsample, ensuring that Pt tip is in contact with the sediments. The electrode should be held in one position without movement. Drift will occur as a rest potential is achieved. This occurs when a relatively stable value (drift <10 mV/min) is achieved (usually within 3 minutes);
* (b) Record the Eh value and temperature at the same time;
* (c) Correct Eh value using temperature correction factors supplied by manufacturer. Some electrodes may not require correction or the associated meter has a setting to account for correction internally. (Check with manufacturer);
* (d) Gently wipe the electrode and remove all sediment prior to insertion into the next sample; and
* (e) Any oily residue on the electrode must be removed prior to further use.
Note: Measured Eh potentials are converted to be relative to the normal hydrogen electrode (NHE) by addition of a potential characteristic for the filling solution used and the sample temperature. For example, the correction factor applied to a potential measured at 20°C with a 4 Mole of Potassium Chloride (M KCl) in saturated Silver Metal / Silver Metal Chloride (Ag/AgCl) reference solution is +204 mV. This value is added to the measured Eh potential (irrespective of whether the potential is positive or negative) to calculate the EhNHE potential. Correction factors for different filling solutions are usually provided by a manufacturer in the electrode instruction manual.
4.8 Biological Sampling
* (a) All sediment should be scraped and rinsed from the grab into pre-cleaned containers when collecting samples for analysis of benthic macrofauna. Save the rinse water as part of the infaunal sample.
Note: Rinse water must be filtered through a minimum 250 μM screen to remove ambient fauna prior to use;
* (b) When sieving biological samples in the field:
* (i) sieve each sediment sample, associated overlying water and rinse water through a 1.0 mm screen. Care must be taken when sieving that fauna is not damaged by abrasion or water pressure. Depending on the volume of the sample, sieving it in batches should be considered;
* (ii) count, identify, photograph and record megafauna then return to the ocean causing it the least amount of harm;
* (iii) retain all coarse gravel and cobble less than 2.5 cm in diameter. Remove attached epifauna and include with infauna separated in the sieved sample;
* (iv) fix the faunal sample in 10% buffered formalin; and
* (v) after four days, rinse the preserved fauna samples on a 0.5 mm screen to remove formalin and then preserve in 70% isopropyl alcohol or ethyl alcohol.
Note: Formalin/rinse water mixture must be contained and appropriately treated prior to disposal; and
* (c) All biological samples shall be:
* (i) counted and taxonomically identified to at least the level of family with all major taxa identified to species; and
* (ii) properly maintained, stored and archived for at least five years after processing.
5. Quality Assurance / Quality Control of Sediment Samples
Quality assurance (QA) and quality control (QC) are essential components of a sampling and monitoring program. Quality assurance can be described as “all those planned and systematic actions necessary to provide adequate confidence that a product or service will satisfy given requirements for quality”.
Quality control can be described as “the operational techniques and activities that are used to fulfill requirements for quality” or “a well-documented plan that ensures results are within a consistent level of error.” There is both a field and lab component to QA/QC. Commercial labs are required to obtain certification from the Canadian Association for Environmental Analytical Laboratories (CAEAL), which requires them to participate in a proficiency testing interlaboratory program. Not all parameters associated with marine sediment analysis are part of this program, and for those, labs must follow a standard quality assurance plan that includes the use of blanks, duplicate samples and reference standards.
5.1 Physical and Chemical QA/QC
* (a) Redox Potential
* (i) ensure the Pt electrode is prepared with filling solution and standardize using recommended redox standards (Section 6);
* (ii) obtain a triplicate measurement of Eh once every 20 samples, or once per batch if fewer than 20 samples are taken; and
* (iii) if potentials do not stabilize (drift >10 mV/min) select a standard wait time (3 minutes recommended) and record values after this period. Note the lack of stabilization and the standard time at which readings were taken.
* (b) Total Free Sulfides (‘S’) in SAOB
* (i) ensure filling solution is added to the Ag+/S= electrode several hours prior to calibration;
* (ii) calibrate electrode using standards and protocols outlined in Section 6; and
* (iii) obtain an additional ‘S’ measurement from a sample once every 20 samples or once per batch if fewer than 20 samples are analyzed.
* (c) Laboratory Analyses
* (i) obtain one additional sediment sub-sample after every batch of 20 samples or once per batch if fewer than 20 samples are taken, for duplicate analyses of TVS, SGS, % moisture (water content) and other applicable parameters;
* (ii) ensure sub-sample is representative of the composite sample, properly homogenized, and large enough for all intended analysis; and
* (iii) upon receiving lab data confirm that:
- 35% Relative Standard Difference for SGS has not been exceeded;
- 20% Relative Standard Difference for TVS has not been exceeded; and
- 40% Relative Standard Difference for metals has not been exceeded.
5.2 Biological QA/QC
There are two options for QA/QC on biological samples:
* (a) Submit QA samples to an expert contract taxonomist. The taxonomist’s lab must have its own QA/QC program;
* (b) Have certified facility staff complete the sampling for macrofauna on site by completing the following steps:
* (i) for every 10 grabs, one additional grab should be collected, split into two equal parts, screened and fauna from both parts preserved. Macrofauna should be enumerated and identified from one half of the sample by facility staff while the other half is submitted to a recognized contract laboratory for similar analysis; and
* (ii) the results obtained by facility staff and the recognized taxonomy lab should be comparable at a similarity level of at least 70%. Results from the contract lab must be reported directly to the Department.
Note: Staff must be certified by an educational institute recognized for expertise in taxonomic identification of macrofauna to the family level.
6. Calibrating the Sulfide (Ag+/S=) Electrode
Ag+/S= electrodes are used to measure free sulfide ions (S= or ‘S’) in sediment samples treated with an antioxidant buffer solution (SAOB). Exposure of sediments to alkaline conditions created by the buffer (pH 12 and greater) may solubilise some fraction of solid phase ‘S’ (pyrite and other metal complexes) in a sample. ‘S’ is therefore operationally defined as total free ‘S’ ions measured in sediment samples buffered with SAOB. Ag+/S= electrodes must be calibrated before the start of each sampling event, and recalibrated a minimum of every 3 hours during an analytical session. The following protocols are generic to the Ag+/S= electrode. Specific models may have slightly different specific requirements and the operator must be familiar with the model prior to calibrating the electrode. If routinely used Ag+/S= combination electrodes should be replaced every couple of years as they tend to become less accurate due to wear and tear. 6.1 Materials and Equipment * (a) Ag+/S= combination electrodes should be used with an appropriate multimeter for measuring electrode potentials. Any millivolt meter with connectors suitable for the electrode can be used; and * (b) The choice of filling solution affects electrode potentials (e.g. if an Orion 96-16BNWP Sureflow combination electrode is used, Optimum Results A Orion 900061 filling solution is the appropriate reference filling solution). The level of the electrode filling solution should always be maintained just below the filler hole in the barrel.
6.2 Summary Table of Required Solutions
Solutions Expiration Period
Sulfide Anti-oxidant Buffer (SAOB) 3 hours
Sulfide Anti-oxidant Buffer excluding L-ascorbic acid 7 days
Stock S= solution 10,000 μM (10-2 M Na2S) 48 hours (cool, dark and air excluded)
Standard S= solution 1,000 μM (10-3 M Na2S) 3 hours
Standard S= solution 100 μM (10-4 M Na2S) 3 hours
Note: The three standards () in the table are recommended for the 3-point calibration. If samples are expected or found to have ‘S’ concentrations below 100 μM then a 10 μM standard should be prepared to create a 3-point calibration series consisting of 10 μM, 100 μM and 1,000 μM. ‘S’ solutions below 100 μM are unstable and these values are also at the minimum sensitivity of most commercially available Ag+/S= combination electrodes.
6.3 Solution Preparation
* (a) Sulfide Anti-Oxidant Buffer (SAOB)
* (i) Materials
- 20.0 g NaOH (sodium hydroxide crystals);
- 17.9 g EDTA (ethylene diamine tetra acetic acid disodium salt dehydrate);
- 8.75 g L-ascorbic acid; and
- de-aerated de-ionized (DI) water.
* (ii) Procedure
- dissolve the NaOH crystals in 250 ml of de-aerated DI water (use a volumetric type flask);
- add EDTA to NaOH solution and swirl until dissolved;
- place solution in plastic screw top jar. Larger volumes can be prepared as necessary provided ratio of reagents is maintained;
- add L-ascorbic acid just prior to the calibration process or analyses of samples. Store in the dark at 4°C; and
- de-aerated DI water must be at similar temperature as that of the sediment to be sampled.
* (b) Stock S= solution 10,000 μM
- (i) Materials
- 0.2402 g Na2S 9H2O (10,000 μM) (pre-weighed and stored in ampoules under nitrogen, argon or helium); and
- de-aerated DI water.
- (ii) Procedure
- working in a fume-hood or well-ventilated area, add the 0.2402 g Na2S 9H2O to a volumetric flask containing 100 mL of de-aerated DI water. Swirl until dissolved. Store refrigerated in dark, air-tight bottle with minimum head space;
- prepare a serial dilution (10,000 μM, 1,000 μM, 100 μM and 10 μM (if required)of ‘S’ standards immediately before calibrating the Ag+/S= combination electrode by transferring 10 mL of the 10,000 μM Na2S stock solution into a volumetric flask and diluting with 90 mL of the de-aerated DI water. This procedure is repeated sequentially using 10 mL of each successive standard and diluting with 90 mL of de-aerated DI water; and
- de-aerated DI water must be at similar temperature as that of the sediment to be sampled.
6.4 Calibration Procedures * (a) Frequency and Handling * (i) the Ag+/S= combination electrode must be recalibrated a minimum of once every 3 hours and each time sampling is initiated at a new farm; * (ii) the tip of the electrode should be gently cleaned using an abrasive strip or detergent solution before each calibration. If difficulty is experienced in obtaining the expected slope for the relationship between mV potential and log ‘S’ concentrations (see subsection (b) below) cleaning may be necessary to remove coatings formed on the electrode tip; and * (iii) the level of the filling solution should also be checked and either topped up or replaced with fresh electrolyte (if the latter option is chosen wait at least 30 minutes for electrode to stabilize prior to using it). * (b) Electrode Calibration * (i) calibration of the Ag+/S= combination electrode is to be carried out by working from the lowest to highest concentration in a standard 3-point series. A fixed volume of a standard is diluted with an equal volume of SAOB (1:1 by volume) immediately before each potential is measured; * (ii) the range of standards used for the 3-point calibration should bracket the range of ‘S’ concentration expected in the sediment samples; * (iii) mV potentials are stored by the multimeter after they become stable (usually between 1 – 3 minutes depending on the make of the meter); * (iv) upon completing the measurement of the third standard instruct the meter to calculate the slope of the concentrations. The theoretical slope is -28 mV for each 10-fold increase in the ‘S’ concentration but because of temperature sensitivities and differences in electrodes the accepted range of values for the slope is between -27 to -33. If the slope is outside * this range repeat the calibration using fresh standards (also check cleanliness of probe, filling solution level, battery function, etc. prior to recalibrating); * (v) following calibration, rinse the electrode with DI water and blot dry before measuring the first sample; and * (vi) upon completion of sediment measurements rinse the electrode and drain filling solution (unless electrode will be used within week).
7. Standardizing the Redox (Pt) Electrode
The Pt electrode is used to measure EhNHE potentials to indicate oxidation/reduction potentials in sediment samples. The measurements do not specify what thermodynamic reactions are involved in forming the potentials measured. The following protocols are generic to the redox (Pt) electrode. Specific models may have slightly different specific requirements and the operator must be familiar with the model prior to standardizing the electrode. 7.1 Materials and Equipment * (a) Any commercially available ion specific electrode (ISE) meter or mV meter with connectors suitable for a combination Pt electrode can be used for EhNHE (redox) measurements; * (b) Redox combination (internal reference) electrodes are refillable or gel-filled and have a BNC connector to allow connection to a mV meter with a similar connector. The refillable option is recommended. It is also recommended that the electrode have a thin disc of Pt (surface area ~1 cm2) rather than a pin at the end of the electrode; * (c) If using a refillable electrode it must be filled with a reference solution. A solution with 4 M KCl saturated with Ag/AgCl is recommended for marine samples; * (d) At least one redox reference solution is required for Pt electrode standardization. These can be purchased from the manufacturer of the electrode; and * (e) Abrasive cleaning strips can be used for cleaning (polishing) the Pt tip of the electrode if coatings are formed during use. A fine powdered detergent can also be used.
7.2 Procedures * (a) The Pt electrode must be rechecked against the standard a minimum of once every three hours and each time sampling is initiated at a new farm; * (b) If using a refillable Pt electrode add filling solution (4 M KCl with saturated Ag/AgCl) at least 24 hours before use; * (c) The reference chamber of the electrode must be completely filled. Check that filling solution is present around the reference junction; * (d) Calibrate the electrode by placing it in a standard solution with a temperature close to that expected in the sediment samples. Record the actual temperature immediately prior to measurements. The mV readings should stabilize rapidly (<30 sec) due to strong oxidation-reduction coupled reactions in a standard solution. Record the potential for comparison with the expected standard value of +220 mV (the potential for a triiodide/iodide redox couple standard at 20°C). The measured value should not exceed + 5 mV. If so, recalibrate electrode following instructions in applicable manual; and * (e) Upon completion of sediment measurements rinse the refillable electrode and drain filling solution (unless electrode will be used within week).
8. Performing Statistical Analyses
For soft sediment sites, statistical analyses are performed for compliance purposes to determine if there are significant differences (p < 0.05) between observed mean sediment total free sulfide (‘S’) and designated compliance standards. Nonparametric tests may be used to assess between-station differences in median values of ‘S’ at facility stations and reference stations.
8.1 Statistical Methods to Determine if Requirements Have Been Met
The following study designs and statistical tests are to be used to determine whether facilities are meeting chemical and biological requirements.
Note: Different statistical software may produce slightly different results. The following are examples only, and were produced using the open-source statistical computing environment.
Example Dataset
Table 1: Mean free sulfide concentrations (μM) at facility and reference stations
| Station | Rep 1 | Rep 2 | Rep 3 | Rep 4 | Rep 5 |
| --- | --- | --- | --- | --- | --- |
| 30 m | 1200 | 1500 | 3000 | 1900 | 800 |
| 125 m | 900 | 700 | 1000 | 600 | 800 |
| Ref 1 | 100 | 150 | 175 | | |
| Ref 2 | 200 | 300 | 250 | | |
| 30 m post-fallow | 800 | 650 | 1100 | 540 | 200 |
(a) Descriptive Statistics (required)
These descriptive statistics provide a summary of the data from each station.
(i) Calculate summary statistics including sample number, mean, median and standard deviation for all stations.
Table 2: Summary Statistics
| Station | number | mean | median | standard deviation |
| --- | --- | --- | --- | --- |
| 30 m | 5 | 1680 | 1500 | 841 |
| 125 m | 5 | 800 | 800 | 158 |
| Ref 1 | 5 | 142 | 150 | 38 |
| Ref 2 | 3 | 250 | 250 | 50 |
(ii) Draw a box-and-whisker plot showing all stations.
Figure 1. Box-and-whisker plot
This figure is an example of a box-and-whisker plot showing the median value (centre of the box), the 25th and 75th percentiles (edges of the box), the minimum and maximum values (end of whiskers) and outliers (dots).
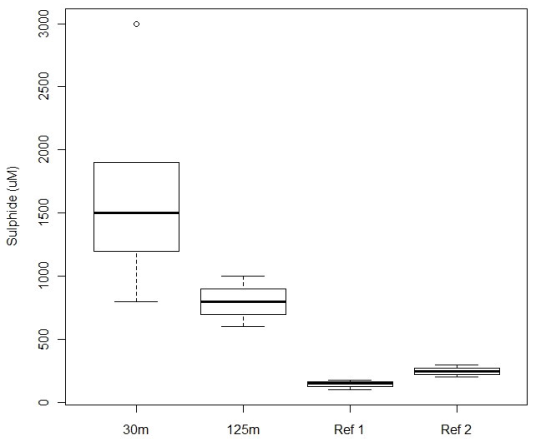 Statistical Test to Determine Compliance (required)
The one-sample t-test is a parametric test to compare measurements from a single station to a single value threshold. It is recognized that with only three to five data points per station, there is not enough information to determine whether all assumptions of the t-test have been met. However, a non-parametric equivalent to this test does not exist. As such, the t-test will be used as the primary method for assessing compliance. The null hypothesis is rejected if the p-value is less than the accepted significance level (α = 0.05).
* (i) Perform a one-sample one-tailed t-test to determine if the mean total free sulfide at the 30 m compliance sampling station is statistically greater than 1300 μM or 4500 μM.
Statistical Test to Determine Compliance (required)
The one-sample t-test is a parametric test to compare measurements from a single station to a single value threshold. It is recognized that with only three to five data points per station, there is not enough information to determine whether all assumptions of the t-test have been met. However, a non-parametric equivalent to this test does not exist. As such, the t-test will be used as the primary method for assessing compliance. The null hypothesis is rejected if the p-value is less than the accepted significance level (α = 0.05).
* (i) Perform a one-sample one-tailed t-test to determine if the mean total free sulfide at the 30 m compliance sampling station is statistically greater than 1300 μM or 4500 μM.
Example:
H0 = the true mean is equal to 1300 μM
HA = the true mean is greater than 1300 μM
Results:
t = 1.011, df = 4, p-value = 0.185
There is no evidence that the mean free sulfide concentration at the 30 m station is significantly greater than the standard of 1300 μM (p > 0.05).
* (ii) Perform a one-sample one-tailed t-test to determine if the mean total free sulfide at the 125 m compliance sampling station is statistically greater than 700 μM.
Example:
H0 = the true mean is equal to 700 μM
HA = the true mean is greater than 700 μM
Results:
t = 1.414, df = 4, p-value = 0.115
There is no evidence that the mean free sulfide concentration at the 125 m station is significantly greater than the standard of 700 μM (p > 0.05).
Annex 9: Monitoring Protocol for Benthic Substrates under Marine Finfish Farms in Newfoundland and Labrador
Objectives
The Monitoring Protocol for Hard Bottom Benthic Substrates under Marine Finfish Farms in Newfoundland and Labrador ( NL) gives guidelines for sampling impacts of marine finfish net pen farm operations on benthic fish habitat to enable evaluation of nutrification effects from deposition of biochemical oxygen demand (BOD) matter (fish feces, uneaten fish feed, and naturally occurring biofouling material). The protocoldescribes the visual benthic sampling procedures conducted at standing biomass necessary to demonstrate compliance with the conditions authorizing the deposit of deleterious substances at an aquaculture facility under the Aquaculture Activities Regulations. It replaces the provisions of the Finfish Fallow Period Monitoring Reporting designed to evaluate the effectiveness of site fallowing as the principal mitigation measure to maintain productive fish habitat. It is applied in circumstances where the nature of the benthic substrate at an aquaculture facility precludes sediment sulfide measurement and when interpretation of the baseline site characterization data normally collected during the aquaculture site licencing process concludes that more than 50% of the lease area is hard bottom composed of rockwall, bedrock, boulders, rubble, cobble, gravel, or hard packed finer substrates. It is expected to be applied at most marine finfish sites in Newfoundland.
Scope
Aquaculture Activities Regulations under the Fisheries Act authorize and establish conditions under which aquaculture operations may deposit drugs, pesticides , and BOD matter during normal operations pursuant to measures in sections 35 and 36 of the Fisheries Act intended to protect fisheries and fish habitat. The regulations are complementary to existing provincial and federal legislative measures and builds upon established siting criteria, mitigation measures, and production management measures. Minimizing BOD matter deposition and avoidance and mitigation of benthic changes at marine cage sites in Newfoundland has historically been accomplished through the application of siting criteria prior to the establishment of sites, adoption of best practices during the production cycle, and establishment of a monitored fallow period between production cycles to allow assimilation of organic deposits through natural processes. These included: * Positioning cages over water depths greater than 30m * Avoiding installations over eelgrass, kelp beds, and other sensitive habitat * Avoiding overlap with existing known commercial fishing grounds * Utilization of feed cameras and daily feed records to minimize excess feed wastage * Application of a minimum one-year fallow period following each production cycle * Waste material and mortalities removal from site * Nets removed and cleaned off-site at centralized facility * Implementation of a "Code of Containment for the Culture of Salmonids in Newfoundland and Labrador" * Equipment removal following production cycle completion * Finfish Fallow Period Monitoring Reporting
Terms and Definitions
aquaculture the cultivation of fish
baseline monitoring sampling of an area of influence which previously has not been used for finfish production
benthic associated with the sea floor
biochemical oxygen demanding matter any organic matter that contributes to the consumption of oxygen that is dissolved in water or sediment
cage floating framework with attached net bag which encloses the fish, and which forms a part of the fish farm
cage area area of seabed directly below the cage
disease event detection of a pathogen or disease that results in a site being quarantined by federal and provincial authorities or as indicated by a veterinarian
environmental monitoring systematic observation, measurement and calculation of the condition of the environment, emission of pollutants, or populations and species, which are necessary for the assessment of the condition of the environment, the development of environment policies, and the planning of environmental protection measures, as well as the control of the effectiveness thereof
fish farm site geographically defined location for aquaculture
hard bottom substrate consisting of bedrock, larger rocks/stones or fixed marine constructions such as wharfs, quays and pipelines
indicator species benthic species that defines a trait or characteristic of the environment or that serves as a measure of the environmental conditions that exist in a given location
macrofauna bottom-dwelling animals retained on a mesh screen of 0.5 mm or 1 mm aperture size
monitoring program set of routine measurements of parameters that describe environmental effects from finfish farms
monitoring standard means the Aquaculture Monitoring Standard, as amended from time to time, that is produced by the Minister and maintained on the Department of Fisheries and Oceans website
soft bottom in relation to a body of water, a bottom or floor that consists of loose particles such as clay, mud, marl, sand, pebbles, gravel, shells or small stones
standing biomass the total biomass held in an aquaculture facility at a given time
threshold value value of a parameter that divides between defined levels of impact in a monitoring program
transect monitoring documentation of qualitative and quantitative changes over a distance
Methodology
Establishing Sampling Stations * Sampling stations should be established at the edge of the cages or containment array and in the immediate vicinity at a set distance from the edge within the predicted or anticipated zone of effect of the facility * Sampling must not be undertaken on any site during any disease outbreak or recovery period as determined in consultation with the NL DFA – Aquatic Animal Health Division. All equipment must be sanitized in accordance with approved biosecurity protocols.
Sites may not be visited under the following circumstances: * Quarantine is in place * During the quarantine period in a Reportable Disease event unless under approved Standard Operating Procedures (SOPs) * Gear or equipment associated with a Reportable Disease event is still present on site * A significant pathogen of provincial concern or an unexplained mortality event has been detected on site * A site has an ongoing diagnostic work up by a Veterinarian and the Veterinarian deems this site as a biosecurity risk
Sites may be visited under the following conditions: * NL DFA – AAHD has been contact prior to visitation and there is no disease event or identified biosecurity risk * Biosecurity Standard Operating Procedures for site visitation are followed * Multiple companies/sites are not visited on the same day * Multiple Bay Management Areas are not visited on the same day
Establishing Reference Stations
The following criteria will be used when establishing a reference station:
* The location is not exposed to BOD matter deposit from the facility or from adjacent facilities as indicated by predictive modeling, historical data, and / or sampling results;
* The depth is within +/- 10m of the range of depths of the sampling stations;
* Topography, seabed type, current and tidal regimes, exogenous influences, sediment grain size and the amount of freshwater runoff influence are representative of the sampling station;
* The reference station should be located in the same contiguous body of water.
Sampling Methodology * Monitoring should be conducted during water and weather conditions that minimize adverse effects on sampling quality and facilitate accurate sampling at stations (for example, at slack tide, during low wind conditions, good visibility) * Sampling should be conducted during the period of actual or predicted maximum daily quantity of feed usage during the production cycle at the facility * Visual sampling should be undertaken in accordance with the Standard Operating Procedure (SOP) for Underwater Video Camera System (2012) * Video may be recorded using a drop (cable) camera. The perimeter of the area under each cage must be videotaped, and as much area under each cage as possible * The video must record the date and be accurately referenced such that an individual viewing the video can appreciate conditions at each sampling location * The video must be well balanced with respect to image focus, clarity, colour balance and lighting * The video must also be accompanied by completed information tables (Appendix 1). Ensure the tables describe the exact site location; images on the tape; depth of water under each cage; and, the camera/video location per cage. See the sample drawings in Appendix 2 * Additional information to be included are as follows:
- DFA AQ number
- Site owner
- Site location
- First year of occupation
- Total years site has been in production
- Cage types, sizes, depths, and numbers in use during production cycle
- Species farmed
- Type of feed used and amount
- Any incidents of escape or significant mortality on the site during the most recently completed production cycle. If yes, identify the cause(s)
-
How mortalities or other farm wastes are disposed
-
For standing biomass monitoring, produce a video recording of conditions at cage edge (0 meters) and continuing at 20m intervals along transects up to a maximum distance of 100m
- Two transects will be carried out from each corner of cage array
- Additional transects will be carried out in center of cage array for sites having more than 9 cages in a row. See Figure 1 in Appendix 2
- Following completion of the first monitoring report, the facility owner and DFO may modify the monitoring transects in subsequent monitoring cycles to target the potential zone of impact
Trout sites
Because trout aquaculture in Newfoundland follows a different production cycle than salmon aquaculture (trout are seasonally raised in an overwintering site and production/growout site), application of this monitoring protocol to the seasonal trout aquaculture sites will be modified as follows: * Benthic monitoring will only be required on sites from which the trout are harvested for market, also known as production sites. * Overwintering (e.g., Roti Bay) sites will not require monitoring using the protocol but will be subject to the carrying capacity and seasonal limits contained in the Roti Bay Management Plan.
Amendments
Any amendments made to this Monitoring Protocol will be done in conjunction with the AAR Working Group.
Appendix 1 – Tables
Table 1: Sample Video Observation Data Template for Hard Bottom Sites(Substrate type)
This table has 18 columns, with headings as follows: Column 1: Station, Column 2: Cage, Column 3: Transect (m), Column 4: Latitude, Column 5: Longitude, Column 6: Depth (m), Column 7: Video quality, Column 8: Figure number, Columns 9 - 17 are under the heading "Substrate", Column 9: Primary/secondary (>50% hard/soft), Columns 10 - 17 are under the sub-heading "Descriptions", Column 10: Rockwall vertical, Column 11: Continuous bedrock, Column 12: Boulders/rubble, Column 13: Cobble, Column 14: Gravel/pebble, Column 15: Sand/mud, Column 16: Organic, Column 17: Floc (% cover), Column 18: Comments and observations.
| Station | Cage | Transect
(m) | Latitude | Longitude | Depth
(m) | Video
Quality | Figure # | Substrate | Comments and observations |
| --- | --- | --- | --- | --- | --- | --- | --- | --- | --- |
| Primary/Secondary
( >50 % hard/soft) | Descriptors |
| Rockwall vertical | Continuous Bedrock | Boulders / rubble | Cobble | Gravel /pebble | Sand /mud | Organic | Floc (% cover) | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
Table 2: Sample Video Observation Data Template for Hard Bottom Sites (Benthic Descriptors)
This table has 20 columns with headings as follows: Column 1: Station, Column 2: Cage, Column 3: Transect (m), Column 4: Latitude, Column 5: Longitude, Column 6: Depth (m), Column 7: Video quality, Column 8: Figure number, Columns 9 - 12 are under the heading "Benthic Indicators", Column 9: Beggiatoa (% cover), Column 10: OPC, Column 11: Type of OPC, Column 12: % of OPC, Columns 13 - 19 are under the heading "Other Benthic Descriptions or Observations", Column 13: Off gas, Column 14: Feed, Column 15: Shell debris, Column 16: Sediment colour, Columns 17 and 18 are under the sub-heading "Flora (% cover)", Column 17: Cor. algae, Column 18: Others, Column 19: Fauna (abundance by group (e.g., Anenomes (2), Starfish (1), Porifera (1) and Column 20: Comments and observations.
| Station | Cage | Transect
(m) | Latitude | Longitude | Depth
(m) | Video
Quality | Figure # | Benthic Indicators | Other Benthic Descriptors or Observations | Comments and observations |
| --- | --- | --- | --- | --- | --- | --- | --- | --- | --- | --- |
| Beggiatoa
(% cover) | OPC | Barren | Off
Gas | Feed | Shell Debris | Sed. Colour | Flora (% cover) | Fauna (abundance by group) |
| Type | % | Cor. Algae | Others | e.g. Anemones (2), Starfish (1), Porifera (1) |
| | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | |
Rhodophyceae (red algae), chlorophyceae (green algae), phaeophytae (brown algae such as kelp)
* crab, soft coral, anemone, sponge, crinoid, urchin, starfish, brittle star, sea cucumber, other (specify)
General Comments on Site: __________________________________________
Completed By: ______
Date Completed: ____ Phone: ________
Appendix 2 – Figures
Sample site layouts and surrounding information
These figures provide guidance on how to diagram the containment array, including global positioning system (GPS) coordinates, for location of where sediment samples should be collected. The labelled diagram must also include location of nearby onshore and offshore infrastructure (wharves, buoys, cabins, etc.).
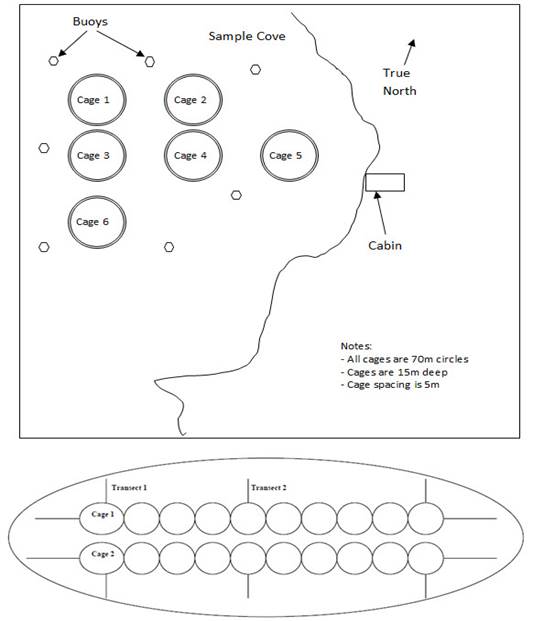
Provide a labelled, dimensioned drawing that illustrates:
- Cages and configuration;
- Transect intervals;
- True North; and
- All onshore and offshore infrastructure (wharves, buoys, cabins, etc.).
Annex 10: Summary of required actions
There are six sections (6, 8, 9, 12, 13, and 16) in the Regulations that require the owner or operator to take specific action. This annex outlines the required actions and provides a schematic summary of the subsequent processes, together with expected timelines. Actions for sections 6 and 13 are related, so the schematic is combined. Actions for 8 and 9 are the same.
Section 6: The owner or operator must notify the Regional Aquaculture Management Office (RAMO) at least 72 hours prior to the deposit of pest control products. Notification must include the following information:
* Product name;
* Date and time of deposit; and
* Place of deposit, using GPS coordinates or their equivalent.
Notification must be made by email. The owner or operator will receive confirmation of receipt of the notification. If the owner or operator does not provide the notification, DFO fishery officers will inform the company that it is in contravention of the AAR. Section 13: The owner or operator must immediately notify a fishery officer if fish morbidity or mortality outside the aquaculture facility is observed (either through visual inspection or by means of underwater cameras) from any part of the facility within 96 hours after the owner or operator has deposited any drug or pest control product referred to in sections 2(a) or (b). Notification must include the following information: * The geographic coordinates of the fish observed; * The estimated number, and if known, the species of the fish observed; * The product name of the drug or pest control product deposited; and * The date of the deposit.
Notification must be made by calling the appropriate toll-free number (see Annex 4). The owner or operator will receive confirmation of receipt of the notification.
In the context of a net pen array, “outside the facility” means outside the net anchors on the bottom and the corresponding area on the surface (see schematic in s.13).
Following a report, DFO will determine, using a science-based triage process, whether a Fisheries Acts.36(6) directive will be issued. Measures under the directive may include taking water, tissue and sediment samples in the affected area, according to the Sampling Guidance for Drugs and Pest Control Products contained in Annex 6.
Figure 1 (below), entitled “Process for pesticide notification and subsequent morbidity/mortality observation,” shows the relationship between the notification of pesticide deposit in section 6(d), the notification of a morbidity or mortality event [s.13(1)] within 96 hours of depositing either a drug or a pest control product, and the steps that must be taken [s.13(2)] if the triage process indicates that the event was caused by the deposit.
Figure 1: Process for pesticide notification and subsequent morbidity/mortality observation
Figure 1 is a flow chart illustrating the process which begins with the owner or operator notifying the RAMO of the intent to deposit 72 hours in advance. The RAMO provides receipt of notification and informs DFO Conservation and Protection (C&P) that the deposit is being made and if there are any concerns. C&P and/or the RAMO may visit the site to verify that the deposit has been made. If the 72-hour notification is not provided, C&P will inform the owner or operator that he is in contravention of the Regulations. Should the owner or operator observe morbidity or mortality among wild fish populations outside the facility, he must notify a DFO fishery officer by calling the appropriate toll-free number listed in Annex 4 and provide geographic coordinates of the event, the estimated number of fish and species, the date of the deposit and name of the product deposited. Confirmation of receipt of notification will be provided. Using the information provided, DFO will perform a triage to examine the cause and gauge the scope of the morbidity/mortality event. Based on the triage result, the RAMO works with the fishery officer to determine whether an investigation is required. If such an investigation is warranted, the fishery officer contacts the owner or operator and issues a s.36(6) directive to sample. The owner or operator must follow the instructions contained in the directive. The owner or operator must submit information regarding all deposits and any morbidity/mortality events in the annual report.
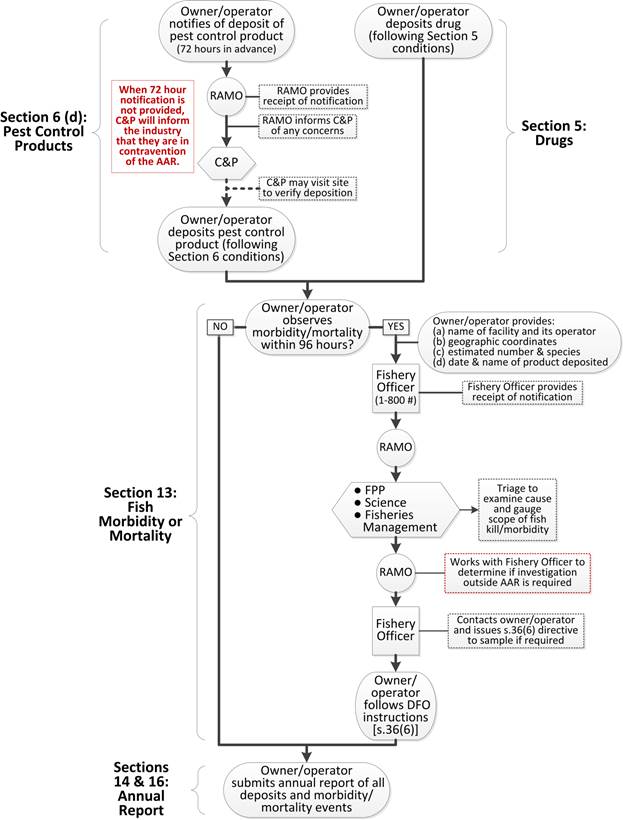 Sections 8 and 9: The owner or operator is required to submit baseline data on new sites (s.8) and for any applications that would result in an increase in the predicted contour of the BOD footprint of an existing site (s.9). Information may be submitted by email or by post; confirmation of receipt will be provided.
* Baseline data for new sites – submit information to the RAMO at least 300 days before making a first deposit of a deleterious substance (drugs, pesticides, or BOD matter). Applications must be complete before consideration can be given.
* Applications affecting contours of footprint – submit information to the RAMO within 30 days after the day on which the application was made.
Sections 8 and 9: The owner or operator is required to submit baseline data on new sites (s.8) and for any applications that would result in an increase in the predicted contour of the BOD footprint of an existing site (s.9). Information may be submitted by email or by post; confirmation of receipt will be provided.
* Baseline data for new sites – submit information to the RAMO at least 300 days before making a first deposit of a deleterious substance (drugs, pesticides, or BOD matter). Applications must be complete before consideration can be given.
* Applications affecting contours of footprint – submit information to the RAMO within 30 days after the day on which the application was made.
Figure 2 (below), entitled “Typical provincial siting process,” is a flow chart showing the start of the process where industry requests siting, the public engagement and data gathering on siting options carried out by the applicant, followed by submission of the final aquaculture licence request. Once the complete request has been received, the 300 days begin. There is a six (6) month siting review period during which DFO has between four and six months to provide its advice to the province for consideration. Once the licence or lease has been granted by the province, the industry has four to six months to install equipment and stock fish.
Figure 2: Typical provincial siting process
 Figure 3 (below), entitled “The 300-day review process,” illustrates the siting review process, starting with the owner/operator collecting the baseline data and submitting the information to both DFO (as required by the AAR) and to the province as required by provincial lease application processes. At this point, the application is reviewed on two tracks: through DFO and through the province. On the DFO level, the RAMO provides confirmation of receipt and sends the package to Fisheries Protection Program, Science, and Fisheries Management. The RDG provides DFO’s complete siting advice to the province which incorporates it into the provincial review of baseline data. Following the conclusion of the provincial review, the owner or operator is informed of the siting decision.
Figure 3: The 300-day review process
Figure 3 (below), entitled “The 300-day review process,” illustrates the siting review process, starting with the owner/operator collecting the baseline data and submitting the information to both DFO (as required by the AAR) and to the province as required by provincial lease application processes. At this point, the application is reviewed on two tracks: through DFO and through the province. On the DFO level, the RAMO provides confirmation of receipt and sends the package to Fisheries Protection Program, Science, and Fisheries Management. The RDG provides DFO’s complete siting advice to the province which incorporates it into the provincial review of baseline data. Following the conclusion of the provincial review, the owner or operator is informed of the siting decision.
Figure 3: The 300-day review process
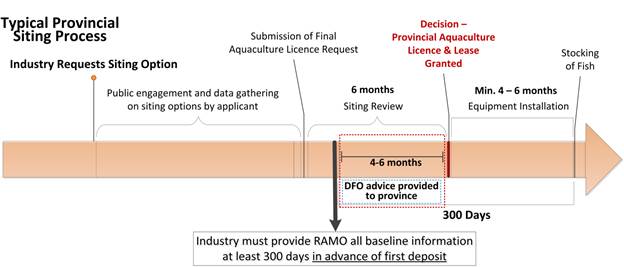 Section 12: Owners and operators are required to monitor sediment sulfide concentrations either through substrate sampling (s.10) or visual monitoring (s.11) to assess the potential impact of organic matter on the benthic environment. If the specified concentration limits are exceeded, the owner or operator must notify the RAMO within 14 days after the day on which the samples of the substrate were taken or the visual monitoring conducted. Information must be submitted by email, and confirmation of receipt will be provided.
Section 16: Anyone holding a provincial/territorial or federal aquaculture licence for a facility that has the potential to deposit deleterious substances or cause serious harm to fish must submit an annual report respecting the operations of each site location during the calendar year. The report is required to be filed by April 1, or three months following the end of the year being reported.
The report must include all the information listed in section 16 of the Regulations (and itemized in the Reporting Template in Annex 1). While using the Reporting Template is not mandatory, it is recommended as it provides a comprehensive checklist of all the information required to be included in the annual report.
Section 12: Owners and operators are required to monitor sediment sulfide concentrations either through substrate sampling (s.10) or visual monitoring (s.11) to assess the potential impact of organic matter on the benthic environment. If the specified concentration limits are exceeded, the owner or operator must notify the RAMO within 14 days after the day on which the samples of the substrate were taken or the visual monitoring conducted. Information must be submitted by email, and confirmation of receipt will be provided.
Section 16: Anyone holding a provincial/territorial or federal aquaculture licence for a facility that has the potential to deposit deleterious substances or cause serious harm to fish must submit an annual report respecting the operations of each site location during the calendar year. The report is required to be filed by April 1, or three months following the end of the year being reported.
The report must include all the information listed in section 16 of the Regulations (and itemized in the Reporting Template in Annex 1). While using the Reporting Template is not mandatory, it is recommended as it provides a comprehensive checklist of all the information required to be included in the annual report.
Annex 11: Glossary of terms
Aboriginal (in relation to a fishery) fish is harvested by an Aboriginal organization or any of its members for the purpose of using the fish as food, for social or ceremonial purposes or for purposes set out in a land claims agreement entered into with the Aboriginal organization.
Act the Fisheries Act.
Aquaculture the cultivation of fish.
Aquaculture facility the location where aquaculture activities are being conducted and encompasses: net pens, walkways, barges, floating structures, tanks, vessels (e.g., well boats), and living accommodations, and any associated lines and anchors. Equipment used in the rearing, handling or transport of fish, or in the operation, cleaning (including off-site net cleaning), disinfection and care of the physical structures themselves (whether on or off lease) is considered to be part of the facility.
Aquaculture licence any of the following: * A lease or licence issued or granted by the Minister under section 7 or 58 of the Act , or its regulations, for the purpose of aquaculture; * A lease granted by the government of a province under subsection 59(1) of the Act for the cultivation of oysters; * A provincial licence or authorization for the operation of an aquaculture facility.
Barren substrate or bare substrate benthic substrate on which there are no visible organisms as a result of organic enrichment from deposits from aquaculture activity; areas where BOD deposits have created hypoxic or anoxic conditions that are inhospitable to organisms including organisms that are indicators of organic enrichment. These are not areas that were bare prior to aquaculture activity.
Bathymetry the measurement of the depths of oceans, seas, or other large bodies of water, as well as the data derived from such measurement, especially as compiled in a topographic map.
Beggiatoa species of bacteria that form visible mats on the seabed surface in areas of organic enrichment.
Benthic associated with the sea floor.
Biochemical oxygen demanding (BOD) matter any organic matter that contributes to the consumption of oxygen that is dissolved in water or sediment.
Commercial (in relation to a fishery) fish is harvested under the authority of a licence for the purpose of sale, trade or barter.
dGPS Differential Global Positioning System is an enhancement to a Global Positioning System that provides improved location accuracy, from the 15-meter nominal GPS accuracy to about 10 cm in case of the best implementations.
Deleterious substance
(a) any substance that, if added to any water, would degrade or alter or form part of a process of degradation or alteration of the quality of that water so that it is rendered or is likely to be rendered deleterious to fish or fish habitat or to the use by man of fish that frequent that water, or
(b) any water that contains a substance in such quantity or concentration, or that has been so treated, processed or changed, by heat or other means, from a natural state that it would, if added to any other water, degrade or alter or form part of a process of degradation or alteration of the quality of that water so that it is rendered or is likely to be rendered deleterious to fish or fish habitat or to the use by man of fish that frequent that water.
Disinfectant an antimicrobial agent capable of destroying pathogenic and potentially pathogenic micro-organisms on environmental surfaces and inanimate objects.
Fish includes (a) parts of fish, (b) shellfish, crustaceans, marine animals and any parts of shellfish, crustaceans or marine animals, and (c) eggs, sperm, spawn, larvae, spat and juvenile stages of fish, shellfish, crustaceans and marine animals.
Fish habitat spawning grounds and any other areas, including nursery, rearing, food supply and migration areas, on which fish depend directly or indirectly in order to carry out their life processes.
Fish that support fish that contribute to the productivity of a fishery.
Hard bottom substrate consisting of bedrock, larger rocks/stones or fixed marine constructions such as wharfs, quays and pipelines.
Mitigation measures to reduce the spatial scale, duration, or intensity of adverse effects to fish and fish habitat that cannot be completely avoided.
Monitoring Standard the Aquaculture Monitoring Standard, as amended from time to time, that is produced by the Minister and maintained on the Department of Fisheries and Oceans web site. This document is incorporated by reference into the AAR.
Morbidity the state of being diseased or unhealthy.
Operation the carrying out of animal husbandry activities (i.e., the management and care of cultured animals).
Owner or operator includes everyone employed by the aquaculture facility or acting as an agent of the owner or operator, whether on a full-time, part-time casual or contract basis.
Oxic state refers to the oxygen content of the sediment. The higher the oxygen content, the greater the biodiversity. The accumulation of organic matter on sediments can reduce the amount of oxygen available for fish species to survive.
Pest any injurious, noxious or troublesome insect, fungus, bacterial organism, virus, weed, rodent or other plant or animal.
Pesticide or pest control product any product, device, organism, substance or thing that is manufactured, represented, sold or used as a means for directly or indirectly controlling, preventing, destroying, mitigating, attracting or repelling any pest. Control products include active ingredients used in the manufacture of end-use products and the end-use products themselves. Includes herbicides, insecticides, fungicides, antimicrobial agents, pool chemicals, microbials, material and wood preservatives, animal and insect repellents, and insect- and rodent-controlling devices.
Recreational (in relation to a fishery) fish is harvested under the authority of a licence for personal use of the fish or for sport.
Serious harm to fish the death of fish or any permanent alteration to, or destruction of, fish habitat. For more detailed information, refer to the Fisheries Protection Policy Statement.
Soft bottom in relation to a body of water, a bottom or floor that consists of loose particles such as clay, mud, marl, sand, pebbles, gravel, shells or small stones.
Standing biomass the total biomass held in an aquaculture facility at a given time.
Date modified: 2018-09-13 xxyy